AP Syllabus focus: ‘For a second‑order reaction, 1/[A] versus time is linear: 1/[A]t − 1/[A]0 = kt.’
Second-order kinetics describes reactions whose concentration change over time follows a distinctive reciprocal pattern. Learning the integrated rate law and how to interpret reciprocal plots lets you recognise second-order behaviour and extract the rate constant from experimental data.
What “second order” means for concentration–time behaviour
Second-order reaction (in A): a reaction whose rate depends on the square of a single reactant concentration, typically , producing a linear relationship between and time.
A key feature is that the concentration of A does not decrease linearly with time, and neither does its logarithm; instead, the reciprocal of concentration increases linearly with time. This is the basis of the reciprocal plot used for identification and analysis.
Second-order integrated rate law (single-reactant form)
The AP Chemistry form you use most often relates the concentration of A at time to the initial concentration of A and the rate constant.
= concentration of reactant A at time (M)
= initial concentration of reactant A (M)
= elapsed time (s, or another consistent time unit)
= second-order rate constant (typically M·s if time is in seconds)
This integrated law is the mathematical statement of the syllabus requirement: for a second-order reaction, plotting versus time produces a straight line.
Reciprocal plots: what to graph and how to read it
Building the linear plot
Reciprocal plot: a graph of (y-axis) versus time (x-axis) used to test for second-order kinetics and determine from the line.
To make and interpret the plot:
Compute for each measured concentration of A (make sure [A] is in M so is in M).
Plot on the y-axis against time on the x-axis.
Assess linearity: second-order behaviour corresponds to an approximately straight line across the measured interval (within experimental uncertainty).
Slope and intercept meaning (link to the integrated law)
From , the line has the form :
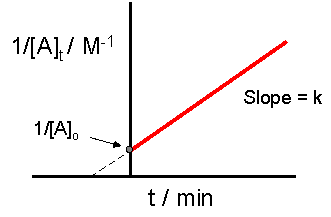
A schematic reciprocal plot used to test for second-order kinetics, with the straight-line trend emphasized. The diagram highlights that the slope of the vs. time line gives the second-order rate constant , while the y-intercept gives . This links the algebraic form directly to experimental graph interpretation. Source
Slope (positive for a typical consumption of A).
y-intercept (the reciprocal of the initial concentration).
A larger k produces a steeper line, meaning increases faster with time and [A] falls more quickly.
Practical constraints and common interpretation checks
When using the reciprocal plot approach, keep these constraints in mind:
Unit consistency matters: if time is recorded in minutes, the slope gives in M·min (not M·s).
Data quality near low concentrations: as [A] becomes small, becomes large, so measurement error in [A] can be magnified on the reciprocal scale.
Linearity is the test: small deviations can arise from mixing delays, temperature drift, or side processes; the goal is an overall linear trend consistent with the integrated law.
Intercept check: extrapolating the best-fit line to should give a y-value close to if the model applies and the initial concentration is reliable.
FAQ
Because the differential rate law leads, after integration, to a linear relationship between $1/[A]$ and $t$.
Mathematically, the curvature in $[A]$ vs $t$ is “unfolded” by the reciprocal transformation.
Look for systematic, one-direction curvature rather than random scatter.
Also check whether early-time points (mixing/temperature equilibration) disproportionately deviate from the best-fit line.
Uncertainty is magnified at low [A]. A small absolute error in [A] can create a large error in $1/[A]$ when [A] is small.
This can make late-time points dominate the visual spread.
Not strictly. The slope still equals $k$, but its units become M$^{-1}$·min$^{-1}$.
Convert only if you need $k$ reported in a particular time unit.
Yes, in special cases (for example, when conditions make the kinetics effectively depend on one changing concentration).
However, confirming why that simplification applies requires additional experimental context beyond the plot itself.
Practice Questions
(2 marks) A reaction is suspected to be second order in A. What plot should be linear if it is second order, and what does the slope represent?
States that a plot of against time is linear (1)
States that the slope equals (the second-order rate constant) (1)
(5 marks) For a reaction involving reactant A, a student plots versus time and obtains a straight line with equation , where is in M and is in seconds. Determine (i) the order with respect to A, (ii) the value and units of , and (iii) .
Identifies second order with respect to A from linear vs time (1)
States equals slope (1)
Calculates (1)
Gives units of as M s (1)
Uses intercept M to get M (1)

