AP Syllabus focus: ‘The same amount of thermal energy does not cause the same temperature change in equal masses with different specific heat capacities.’
Materials respond differently to heating because temperature reflects average particle kinetic energy, while added energy can be distributed among many microscopic “storage” pathways. Specific heat capacity quantifies this material-dependent response.
Core idea: same energy in, different temperature change out
When two objects of equal mass absorb the same amount of thermal energy, they generally show different temperature changes because their particles require different amounts of energy to raise average kinetic energy by 1°C.
Specific heat capacity controls the temperature response
A material with a high specific heat capacity needs more energy to produce a given temperature increase, so it warms (and cools) more slowly. A material with a low specific heat capacity warms more quickly for the same energy input.
Specific heat capacity (c): the amount of heat energy required to raise the temperature of 1 gram of a substance by 1°C (units: J g °C).
This definition is operational: it directly describes how strongly a substance “resists” temperature change when energy is transferred as heat.
Quantitative relationship used to compare materials
The key relationship links thermal energy transfer, mass, specific heat capacity, and temperature change.
= heat transferred (J)
= mass of substance (g)
= specific heat capacity (J g °C)
= temperature change, (°C)
For fixed and equal , the equation implies is inversely proportional to : larger means smaller temperature change.
Particle-level reasons c differs among substances
Different substances partition absorbed energy differently at the microscopic level, so the same energy input does not translate into the same rise in average translational kinetic energy (temperature).
Energy distribution among microscopic “modes”
Added thermal energy can be distributed into:
Translational motion (directly increases temperature)
Rotational and vibrational motion (internal motion that may increase without as large a temperature rise)
Collective motions in solids (lattice vibrations/phonon-like behaviour)
Substances with more accessible ways to store energy internally tend to have higher specific heat capacities, because more energy is needed before temperature rises noticeably.
Interparticle interactions and structure matter
How strongly particles interact affects how energy is “taken up”:
In many solids, energy is readily shared through the lattice; the detailed structure and bonding influence how much energy changes particle motion per degree.
In liquids, close packing and interactions can provide additional pathways for energy storage beyond simple speeding up of particles.
Why water is a classic “slow warmer”
Water’s relatively high is often attributed to strong intermolecular attractions and many available microscopic motions, so added energy is spread out rather than immediately appearing as a large temperature increase. In contrast, many metals have comparatively low and show larger temperature changes for the same heating.
What “equal masses” and “same energy” really imply experimentally
To compare materials fairly, you must control:
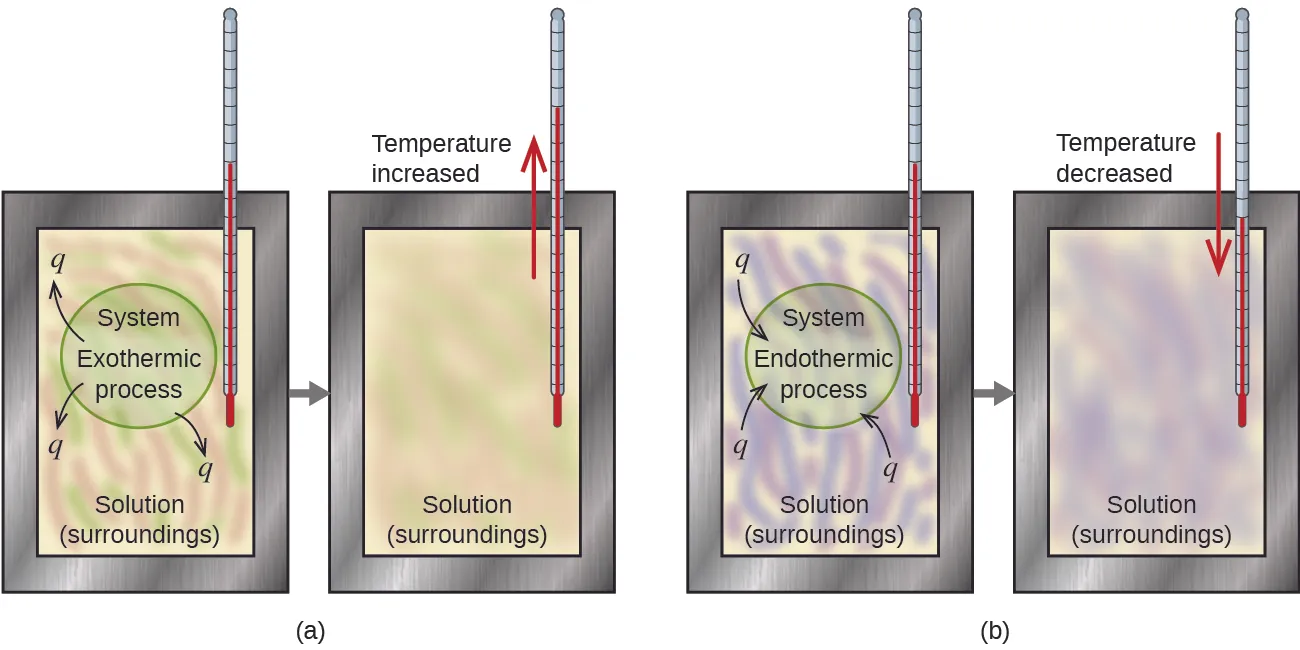
Two schematic calorimetry setups contrast exothermic and endothermic processes by showing the direction of heat flow () and the resulting temperature change in the solution. The diagram reinforces that the thermometer reading is an observable proxy for energy transfer under (ideally) insulated conditions. This is the conceptual backbone for interpreting measurements when applying . Source
Mass: equal grams of each substance
Energy input: same heat transferred into each sample (not merely heating for the same time unless the heater power and losses are controlled)
Heat losses: insulation and consistent setup, since losses reduce the effective delivered to the sample
Because temperature is a measure of average kinetic energy, observing different values for equal and is direct evidence that material-dependent specific heat capacity governs how thermal energy translates into temperature change.
FAQ
Often it is treated as constant over small temperature ranges, but it can vary with temperature and physical state.
They usually conduct heat away from your skin faster, so your skin cools more quickly even though the initial temperatures match.
Impurities can change composition and structure, shifting $c$; mixtures often have an effective $c$ between their components.
At constant pressure, some energy contributes to expansion work; at constant volume it cannot, so the measured heat capacity differs.
Heat loss to the surroundings, poor mixing, temperature probe lag, and unaccounted heat absorbed by the container can mask real differences.
Practice Questions
(2 marks) Two 50.0 g samples, A and B, each absorb J of heat. Sample A increases by C, sample B by C. Which sample has the higher specific heat capacity, and why?
Identifies B has higher specific heat capacity (1)
Explains: for same and , smaller implies larger using or equivalent reasoning (1)
(5 marks) Explain, using particle-level ideas and the relationship , why equal masses of different materials can show different temperature rises when supplied with the same thermal energy. Your answer should refer to how energy is distributed within substances.
States that for fixed and , depends on (1)
Links higher to smaller temperature change / lower to larger temperature change (1)
Describes temperature as average kinetic energy of particles (1)
Explains energy can be stored in different microscopic modes (e.g. vibration/rotation/lattice motion), not only translational motion (1)
Connects differences in bonding/structure/interactions to different energy storage pathways and thus different values (1)

