AP Syllabus focus: ‘In dissolution calorimetry, a temperature increase indicates an exothermic process, while a temperature decrease indicates an endothermic process.’
Dissolution calorimetry uses measured temperature changes when a solute dissolves to infer the direction of energy flow. Correct interpretation depends on defining the system and surroundings and using consistent heat-sign conventions.
What dissolution calorimetry measures
System, surroundings, and what the thermometer “sees”
Dissolution calorimetry: An experimental method that determines heat transfer during dissolving by monitoring the temperature change of the solution (and calorimeter) after a solute is added to a solvent.
In most AP-style setups, the system is the dissolving process (solute + solvent interactions), while the surroundings are the solution water and the calorimeter hardware that the thermometer is in contact with.

Photograph of a typical coffee-cup (polystyrene) calorimeter used for solution calorimetry experiments. The nested insulating cups and a lid reduce heat exchange with the external environment, while the thermometer measures the temperature of the solution (the surroundings). This helps anchor the “what the thermometer sees” idea: it directly tracks the surroundings’ , not the system’s enthalpy change. Source
Inferring energy flow from temperature change
The key interpretation is qualitative:
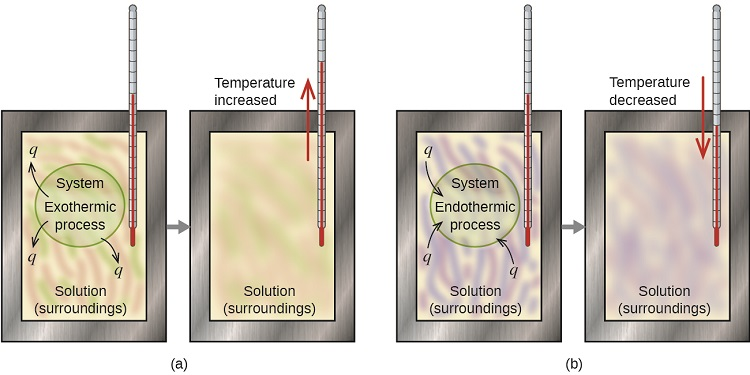
Diagram comparing exothermic vs. endothermic processes in a (constant-pressure) coffee-cup-style calorimetry setup. It explicitly links the observed direction of temperature change in the solution (surroundings) to the sign of heat flow for the system: exothermic processes warm the solution and have , while endothermic processes cool the solution and have . Source
Temperature increases (): heat flowed into the surroundings, so the dissolving process released heat → exothermic.
Temperature decreases (): heat flowed out of the surroundings, so the dissolving process absorbed heat → endothermic.
Exothermic process: A process in which the system releases heat to the surroundings, corresponding to a negative enthalpy change () under constant-pressure conditions.
Because the thermometer measures the surroundings’ temperature, a warmer solution indicates the surroundings gained energy, which must have come from the system.
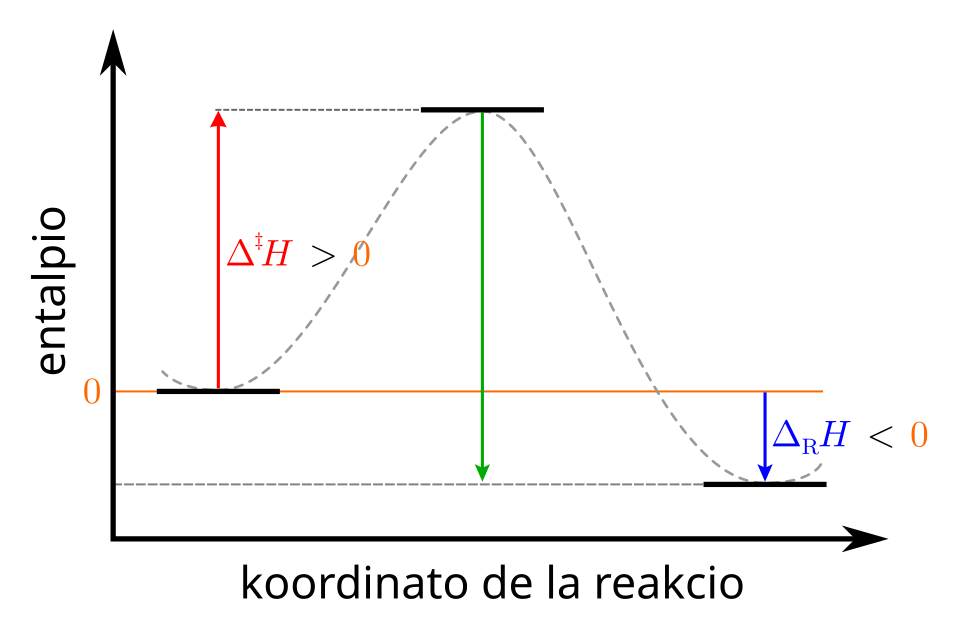
Enthalpy (energy) profile diagram for an exothermic process, showing products at lower enthalpy than reactants. The vertical drop represents the negative enthalpy change, , consistent with heat being released from the system to the surroundings. This provides an energy-level view that pairs naturally with the observed sign of in dissolution calorimetry. Source
Endothermic process: A process in which the system absorbs heat from the surroundings, corresponding to a positive enthalpy change () under constant-pressure conditions.
Energy bookkeeping used in dissolution calorimetry
Heat gained/lost by the solution
To connect temperature change to heat transfer, you typically treat the solution as the part of the surroundings whose temperature changes measurably.
= heat gained by (or lost from) the solution, J
= mass of the solution (often approximated as mass of water + solute), g
= specific heat capacity of the solution (often approximated as ), J g °C
= , °C
This equation quantifies what the temperature change implies about the surroundings’ heat change, not directly the system’s.
Connecting solution heat to dissolution heat
In an idealised coffee-cup calorimeter, the total heat exchange between system and surroundings is conserved, so the system’s heat is the negative of the surroundings’ heat.
= heat absorbed by (or released from) the dissolving process (system), J
= heat gained by (or lost from) the solution (surroundings), J
Therefore:
If , then and → exothermic dissolution.
If , then and → endothermic dissolution.
Practical interpretation and common experimental issues
What “temperature increase/decrease” really means in practice
A measured temperature change reflects several competing effects:
Heat from the dissolution process itself
Heat absorbed by the calorimeter cup/lid/thermometer
Heat exchange with the air (especially if mixing is slow or the cup is open)
Because of these, measured is often a slight underestimate of the true magnitude (warming looks less warm; cooling looks less cool).
Data handling habits that improve inference (without changing the core rule)
Record temperature frequently before and after adding solute to establish a stable baseline.
Stir consistently to reduce temperature gradients so the thermometer reflects the bulk solution.
If temperature drifts over time, use the temperature at the actual mixing time (or an extrapolated value) rather than a late-time reading.
These practices do not change the syllabus rule; they help ensure the observed direction of correctly reflects the direction of heat flow.
FAQ
Fast local dissolving near the solute can create hot/cold spots, then mixing equalises the temperature.
Heat exchange with the cup and air can also pull the temperature back towards room temperature after the main dissolving event.
Many lab protocols use the mass of the whole solution (water + solute) because that is what is changing temperature.
If only the water mass is used, the calculated heat magnitude will be slightly smaller; your teacher’s stated convention should be followed consistently.
If the apparatus is not well insulated or the calorimeter is known to absorb a noticeable amount of heat, a separate calorimeter term may be needed.
This is commonly handled with a “calorimeter constant” determined by calibration, then adding that heat term to the surroundings’ total.
The measured $\Delta T$ depends on both the heat released/absorbed and the total heat capacity of the surroundings.
Changing the amount of water changes the mass that must be warmed/cooled, so the same per-mole enthalpy can produce a smaller or larger temperature change.
Evaporation can occur during mixing, which absorbs heat and can mimic endothermic behaviour.
For gases, incomplete dissolution and slow equilibration can make the recorded temperature change lag behind the actual heat transfer timing.
Practice Questions
(2 marks) When of an ionic solid is added to water in a polystyrene cup, the temperature rises from to . State whether the dissolution is exothermic or endothermic, and justify your answer.
Exothermic (1)
Temperature increase means heat transferred to surroundings / solution gained heat so system released heat (1)
(5 marks) A student dissolves of in of water in a coffee-cup calorimeter. The temperature changes from to . Assume and mass of solution . (a) Calculate . (b) Determine the sign of . (c) Calculate in for (molar mass ).
(a) (1)
(a) (1)
(b) , so positive (1)
(c) Moles (1)
(c) (1)

