AP Syllabus focus: ‘Specific heat capacity and molar heat capacity are both used in energy calculations.’
Heat capacity values let you connect a measured temperature change to an energy transfer. AP Chemistry commonly uses two related constants—one based on mass and one based on amount in moles—so choosing the correct one is essential.
Two heat-capacity quantities you must distinguish
Specific heat capacity (mass-based)
Specific heat capacity, : the heat energy required to raise the temperature of 1 g of a substance by 1°C (or 1 K); units typically J g °C (or J g K).
Specific heat capacity is most convenient when the problem gives the mass of a sample (common in calorimetry with liquids and solids).
Molar heat capacity (mole-based)
Molar heat capacity, : the heat energy required to raise the temperature of 1 mol of a substance by 1°C (or 1 K); units typically J mol °C (or J mol K).
Molar heat capacity is most convenient when the problem describes amounts in moles, which aligns naturally with chemical equations and stoichiometry.
Using each quantity in energy calculations
Temperature changes depend on energy transferred as heat and how much substance you have, so the same physical idea can be written in two parallel forms.
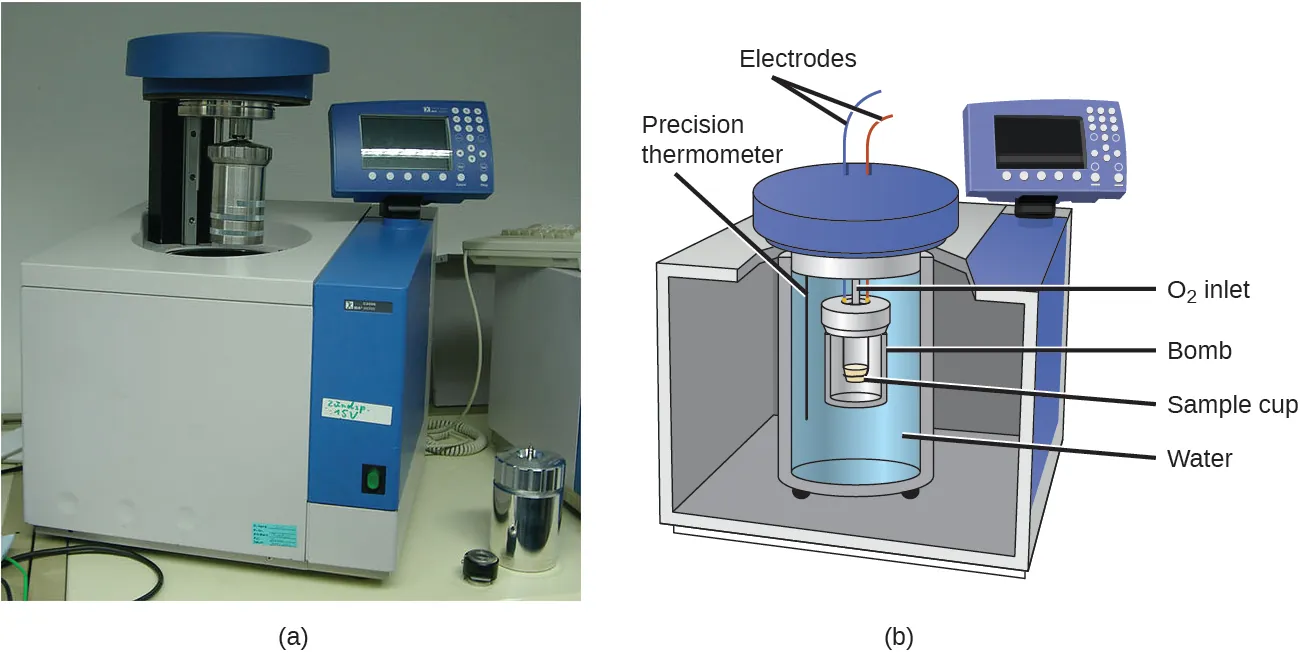
A bomb calorimeter diagram (with accompanying photo) identifying key components like the sealed “bomb,” surrounding water bath, electrodes/ignition, and precision thermometer. The figure illustrates how an exothermic reaction’s released energy is captured as a measurable of the calorimeter/water system, enabling calorimetric energy calculations. Source
= heat absorbed or released by the sample (J)
= mass of sample (g)
= specific heat capacity (J g °C)
= (°C or K)
This form is best when you measure or are given grams of material (for example, a metal sample or a mass of water).
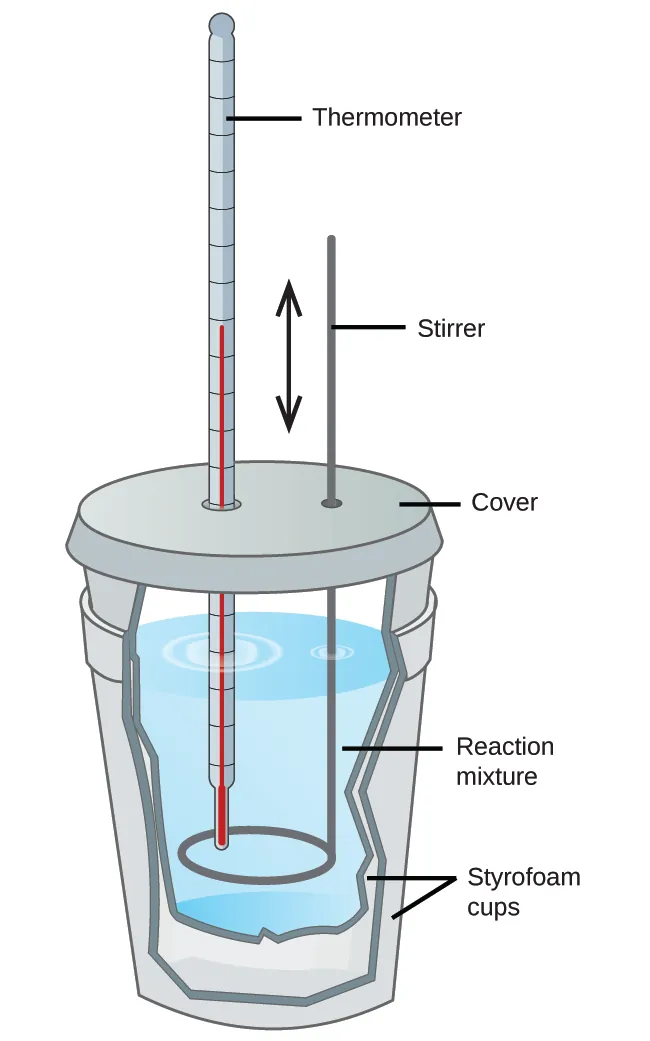
A schematic of a simple coffee-cup calorimeter showing the insulated Styrofoam cups, thermometer, stirrer, and reaction mixture. This is the standard setup where you measure a solution’s and use to calculate heat absorbed or released by the surroundings. Source
= heat absorbed or released by the sample (J)
= amount of substance (mol)
= molar heat capacity (J mol °C)
= (°C or K)
This form is best when the amount is tracked in moles, such as when a temperature change is linked to a reaction mixture and you want energy per chemical amount.
Converting between and
For a pure substance, the two constants describe the same thermal “resistance,” just normalised differently. They are connected by molar mass.
If is molar mass in g mol, then:
This conversion is especially useful when a data source provides one form but the question setup naturally suggests the other (mass given vs moles given).
How to decide which heat capacity to use
Use the quantity that matches what you can measure directly and what the question emphasises.
Choose when:
mass is given in g (or can be found easily)
the substance is acting as the calorimeter fluid (often an aqueous solution is approximated using a single value)
Choose when:
amounts are given in mol
you want to compare substances on a per-mole basis (e.g., “per mole of substance warmed”)
A common pitfall is mixing bases: plugging moles into or grams into creates unit inconsistency and incorrect energy values.
Interpreting the magnitude (what big vs small means)
Both constants indicate how strongly temperature responds to added/removed heat.
Large (or large ) means:
more energy is needed for the same
the substance’s temperature changes less for a given heat transfer
Small (or small ) means:
less energy is needed for the same
the substance’s temperature changes more for a given heat transfer
Because scales with molar mass, two substances can have similar values but different values, or vice versa, depending on how much mass is contained in one mole.
FAQ
When the problem is framed in moles (e.g., linked to a chemical amount) but the heat capacity provided is mass-based.
Yes for temperature changes: $\Delta T$ in K equals $\Delta T$ in $^\circ$C because the size of one degree is the same.
Because $C_m=cM$. A larger molar mass increases $C_m$ even if $c$ is modest, shifting comparisons on a per-mole basis.
No. Sample heat capacity depends on how much you have: $C_{\text{sample}} = mc$ or $C_{\text{sample}} = nC_m$ (units J $^\circ$C$^{-1}$).
They can vary with temperature, purity, and composition; using an accepted value (often for dilute aqueous solutions) introduces systematic uncertainty.
Practice Questions
State what is meant by (i) specific heat capacity and (ii) molar heat capacity, including suitable units. (3 marks)
Correct definition of specific heat capacity: energy to raise temperature of 1 g by 1°C (or 1 K) (1)
Correct unit for : J g °C (or J g K) (1)
Correct definition + unit idea for molar heat capacity: energy to raise temperature of 1 mol by 1°C (or 1 K), unit J mol °C (or J mol K) (1)
A pure substance has and molar mass .
(a) Determine with units. (2 marks)
(b) Write the appropriate expression to calculate needed to raise the temperature of mol of the substance by C, and state why this form is appropriate. (3 marks)
2a)
Uses (1)
Correct value with units: (allow suitable sig figs) (1)
2b)
Correct expression: (1)
Substitutes: (1)
Justification: amount given in moles / molar heat capacity is per mole so matches the data (1)

