AP Syllabus focus: ‘Chemical systems change energy through heating/cooling, phase transitions, and chemical reactions.’
Chemical systems can gain or lose energy in three distinct ways. Recognizing which pathway is occurring helps you interpret temperature data, particle-level changes, and what a calorimetry measurement is actually capturing.
The three pathways: what can change in a chemical system?
A system’s energy can change by shifting how energy is stored among particles. In AP Chemistry, track these three pathways:
Heating/cooling (temperature change)
Phase transitions (state change at constant temperature)
Chemical reactions (composition change)
A single real process may involve more than one pathway at the same time, but each pathway has a characteristic “signature” you can identify.
Pathway 1: Heating and cooling (temperature changes)
Heating/cooling changes the average kinetic energy of particles, which shows up as a temperature change.
Heating: energy is transferred into the system, increasing particle motion and temperature.
Cooling: energy is transferred out of the system, decreasing particle motion and temperature.
Calorimetry often focuses on this pathway because temperature is easy to measure and can be linked to energy transfer.
= heat transferred to/from the sample, in J
= mass of the sample, in g
= specific heat capacity, in
= , in (or K)
Interpreting the sign conceptually:
If the sample’s temperature increases, the sample gained energy by heating.
If the sample’s temperature decreases, the sample lost energy by cooling.
Pathway 2: Phase transitions (state changes)
A phase transition changes how particles are arranged (and how strongly they interact) without changing the chemical identity of the substance.
Key observable feature:
Temperature can remain constant during the phase change, even though energy is still being transferred.
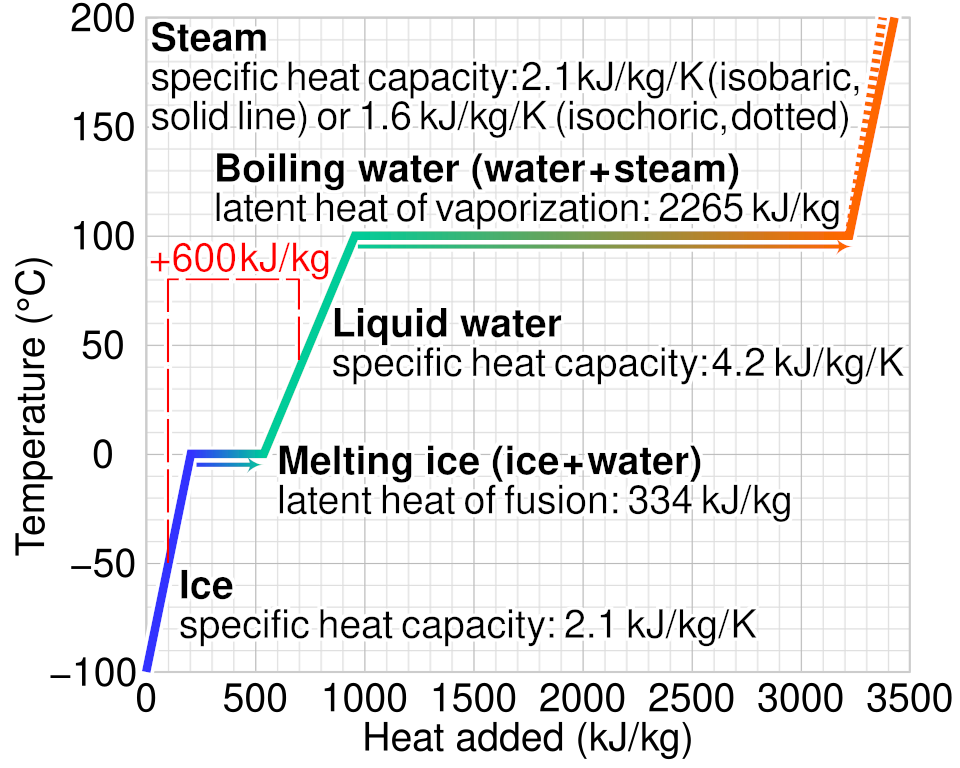
Heating curve for water showing temperature versus heat added, with sloped regions where added energy increases average kinetic energy (temperature rises) and flat regions where added energy drives a phase change at constant temperature. The plateaus correspond to latent heat during melting/freezing and boiling/condensing, where potential energy changes dominate. This is a clean visual cue for distinguishing heating/cooling from phase transitions in experimental temperature data. Source
Particle-level interpretation:
Energy transferred during a phase change primarily changes potential energy (interparticle attractions and spacing), not average kinetic energy.
When attractions are being overcome (like moving toward a less condensed state), energy goes into separating particles.
When attractions are forming more extensively (like moving toward a more condensed state), energy is released as particles come closer and interact more strongly.
This is why a system can absorb or release energy even if a thermometer does not show a temperature change during the transition itself.
Pathway 3: Chemical reactions (chemical composition changes)
A chemical reaction changes which substances are present, meaning bonds and/or interactions are reorganized. This changes how energy is stored as chemical potential energy.
What you can observe experimentally:
The reaction mixture may warm up or cool down because energy is exchanged between the reacting system and its surroundings.
Temperature change is not “the reaction energy” directly; it reflects energy transferred as heating/cooling to surrounding materials (solution, calorimeter, air), which is then used to infer the reaction’s energy change.
Particle-level interpretation:
Some energy is required to disrupt existing attractions (including bonds).
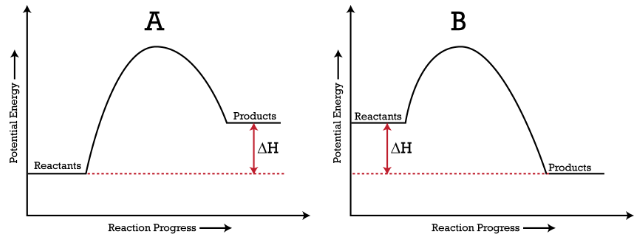
Potential energy (reaction progress) diagrams comparing endothermic and exothermic reactions, with reactants, products, and labeled. The curves emphasize that reactions require an activation-energy barrier, while the vertical difference between reactants and products represents the net enthalpy change. This reinforces that temperature changes in calorimetry are indirect evidence of energy transfer tied to the reaction’s overall energetics. Source
Some energy is released when new attractions form.
The balance between these determines whether, overall, the reacting system absorbs energy or releases it.
Distinguishing the pathways in lab observations
When interpreting experimental data, separate what is happening physically:
If temperature changes smoothly with no plateau: heating/cooling is occurring.
If temperature holds steady while energy transfer continues: a phase transition is occurring.
If new substances form (gas production, precipitate formation, colour change, odour change) and temperature shifts: a chemical reaction pathway is involved, often alongside heating/cooling of the surroundings.
In many setups (especially aqueous reactions), the measured temperature change most directly tracks the heating/cooling pathway of the solution and apparatus, while the chemical reaction pathway is the underlying cause of that heat exchange.
FAQ
Look for mixed signals: temperature change (heating/cooling) alongside evidence of a reaction (new substances) or a phase change (slurry, melting/freezing).
Clues include delayed temperature response, incomplete dissolution, or visible phase coexistence.
Stirring reduces temperature gradients so the thermometer reflects the bulk average kinetic energy more accurately.
Without stirring, local hot/cold spots can make the pathway appear inconsistent or shift the apparent timing of a plateau.
The thermometer measures the solution’s temperature, so it directly tracks heating/cooling of the surroundings.
The reaction’s energy change is inferred because it drives that heat exchange, but it is not measured as a separate “reaction temperature”.
Phase transitions typically show a stable temperature plateau with visible coexistence of phases (e.g., solid + liquid).
Slow reactions may show changing appearance (e.g., gradual colour change) without a sharp plateau, and may depend strongly on concentration.
Heating/cooling is often tracked in J via temperature and heat capacity, while phase and reaction changes are often reported per amount of substance.
Being explicit about “per gram” vs “per mole” prevents mixing pathway descriptions with inconsistent scaling.
Practice Questions
(2 marks) State the three pathways by which chemical systems change energy.
Heating/cooling (1)
Phase transitions (1)
Chemical reactions (1)
(5 marks) A student observes that a substance is being supplied energy at a steady rate. The temperature rises, then remains constant for several minutes, then rises again. Explain, in terms of the three energy-change pathways and particle behaviour, what is happening during each region.
Temperature-rise regions correspond to heating: average kinetic energy increasing (1)
Constant-temperature region corresponds to a phase transition (1)
During phase transition, energy changes potential energy/interparticle attractions rather than average kinetic energy (1)
Particle-level description: separation/arrangement changes while substance identity remains the same (1)
Correctly identifies that the three pathways are distinct and links the observation to heating/cooling and phase change (1)

