AP Syllabus focus: ‘Heating increases a system’s energy, while cooling decreases a system’s energy.’
Heating and cooling describe energy transfer between a chosen system and its surroundings. In AP Chemistry, you connect observed temperature changes to the direction of heat flow and the sign of the system’s energy change.
Heating vs. Cooling: What Changes in the System?
A system is the part of the universe you choose to study (for example, a beaker of solution), while everything else is the surroundings.
System — the matter being studied; energy changes are tracked for the system relative to the surroundings.
When a system is heated, energy enters the system from the surroundings. When a system is cooled, energy leaves the system and is transferred to the surroundings. This is the meaning of the syllabus statement: heating increases a system’s energy, and cooling decreases a system’s energy.
Particle-Level Interpretation
Temperature is linked to particle motion. Heating typically increases the system’s average particle kinetic energy (faster motion), while cooling decreases it (slower motion).
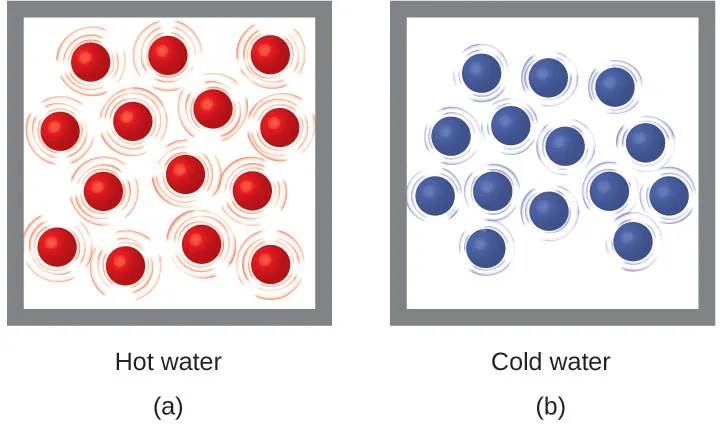
Particle model comparing a hot liquid (faster molecular motion) to a cold liquid (slower motion). The visual links macroscopic temperature to microscopic average kinetic energy, clarifying why heating generally increases particle motion while cooling reduces it. This supports interpreting thermometer readings as evidence of changing molecular kinetic energy. Source
In many lab situations (like solutions in a coffee-cup calorimeter), this kinetic-energy change is what you detect as a temperature rise or drop.
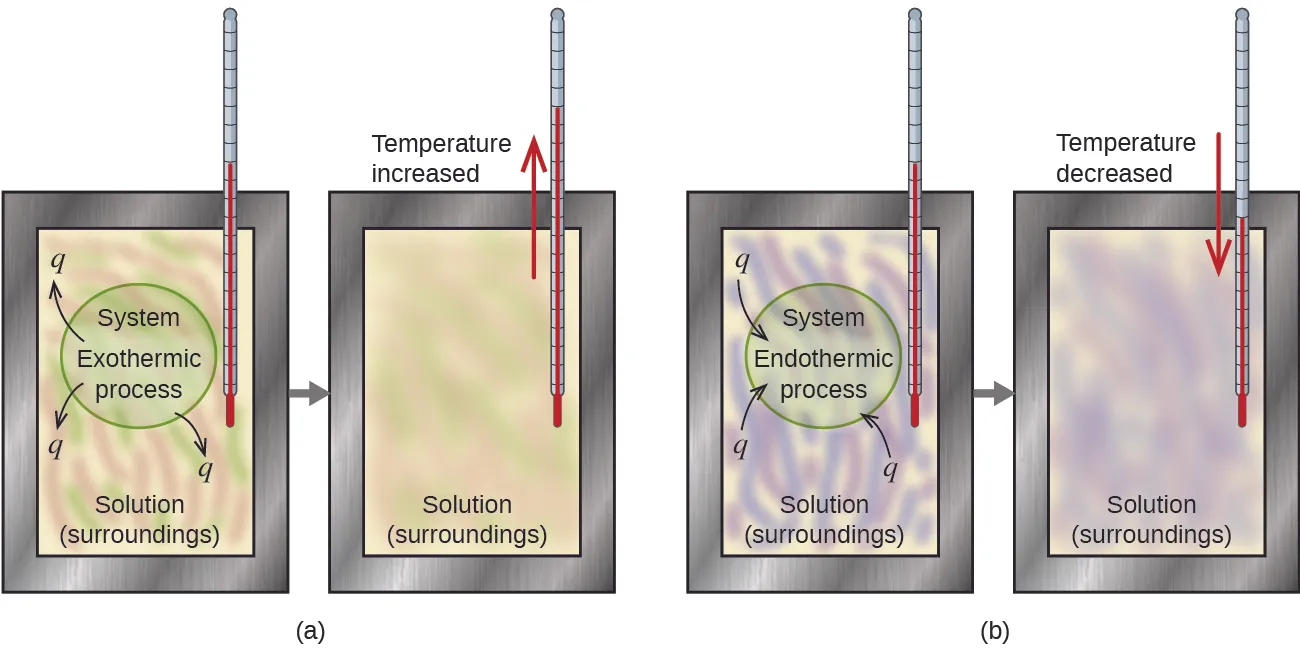
Coffee-cup calorimetry schematic showing how the solution’s temperature change reveals the direction of heat flow. In an exothermic process, heat leaves the system and the solution temperature rises; in an endothermic process, heat enters the system and the solution temperature falls. The diagram also reinforces the AP convention that is negative for exothermic and positive for endothermic processes. Source
Heat Flow and Sign Conventions (What “Energy Increases/Decreases” Means)
In calorimetry-style problems, energy transferred due to a temperature difference is tracked as heat, symbol .
Heat () — energy transferred between a system and surroundings because of a temperature difference.
By convention in AP Chemistry:
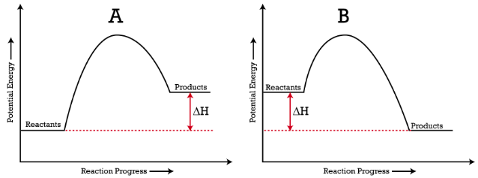
Potential-energy diagrams for endothermic and exothermic reactions plotted versus reaction progress. In the endothermic case, products lie higher than reactants and ; in the exothermic case, products lie lower than reactants and . This connects sign conventions to an energy landscape students commonly use in AP thermochemistry. Source
If the system absorbs heat (heating), then and the system’s energy increases.
If the system releases heat (cooling), then and the system’s energy decreases.
This sign convention is about the system, so always identify the system explicitly before assigning signs.
In typical insulated calorimetry reasoning, the heat change of the surroundings is equal in magnitude and opposite in sign to the heat change of the system (energy transferred doesn’t disappear; it moves).
Connecting Heating/Cooling to Measurable Temperature Change
For many heating/cooling processes without additional complications, the heat transferred is related to temperature change by:
= heat absorbed by the sample (J)
= mass of the sample (g)
= specific heat capacity (J g °C)
= (°C)
A key interpretation is that the sign of matches the sign of for that measured object:
If , the object warmed, so it absorbed heat (heating, for that object).
If , the object cooled, so it released heat (cooling, for that object).
Common “System vs. Surroundings” Temperature Patterns
In many lab setups, you infer what happened to the system by watching the surroundings (often the water/solution you measure):
If the solution temperature increases, the solution gained heat; something else (often the system of interest) must have lost heat to it.
If the solution temperature decreases, the solution lost heat; something else must have gained heat from it.
The critical skill is to keep the perspective consistent: the observed thermometer reading belongs to a specific material, and the sign of you report depends on whether that material is defined as the system.
Avoiding Common Sign and Interpretation Errors
Don’t assign until you state what the system is.
Don’t mix up “temperature is higher” with “contains more energy”: energy change depends on mass and heat capacity, not temperature alone.
If the measured object cools (), its is negative even if another part of the setup is gaining energy at the same time.
FAQ
Pick the matter whose energy change the question cares about (often stated explicitly).
If not explicit, typical choices are:
the reacting chemicals (system)
the solution/water in the cup (surroundings used as a “thermometer”)
State your choice before assigning signs.
It means the particular object whose temperature you measured cooled and therefore had $q<0$.
Another object in contact can simultaneously be heating up with $q>0$, depending on what you defined as the system.
“Heating” refers to heat transfer direction, not the source.
If the system transfers energy to the surroundings (for any reason), the surroundings are heated (their energy increases), even with no external heater.
Heat exchange with the external environment can distort the sign and size of inferred $q$.
Common causes:
poor insulation
delayed temperature reading (missing the true peak/minimum)
evaporation or splashing changing the effective mass
Report $q$ in joules (J) with a sign that matches your defined system.
Match significant figures to the limiting measured quantity (often $\Delta T$), and keep units consistent (e.g., $c$ in J g$^{-1}$ °C$^{-1}$ implies mass in g and $\Delta T$ in °C).
Practice Questions
(2 marks) A student defines the system as a metal sample. The sample’s temperature decreases when placed in contact with water. State the sign of and whether the system’s energy increases or decreases.
(1)
System energy decreases (1)
(5 marks) In an insulated setup, a student observes that the temperature of the surrounding water increases by C after contact with a reaction vessel. The student defines the reaction mixture as the system. (a) State the sign of . (1) (b) State the sign of . (1) (c) Explain, in terms of heating vs cooling and energy transfer, what happened to the system. (3)
(a) (1)
(b) (1)
(c) Any three:
Water warmed, so it absorbed heat (1)
Heat must have come from the system in an insulated setup (1)
The system cooled/released heat, so its energy decreased (1)

