AP Syllabus focus:
‘Compare reservoirs that store carbon compounds for long periods with those that store carbon for relatively short periods.’
Carbon moves through Earth systems by being stored in “reservoirs” for different lengths of time. Understanding short-term versus long-term carbon storage helps explain ecosystem carbon availability and why some carbon pools change quickly while others change slowly.
Core idea: reservoirs and storage time
Carbon reservoir: a physical pool that stores carbon-containing compounds (for example, biomass, soil organic matter, seawater, or carbonate rock).
Reservoirs are compared mainly by storage time (how long carbon remains before moving elsewhere) and by how rapidly carbon can be exchanged with other reservoirs.
Residence time: the average time a molecule of carbon stays in a reservoir before leaving, often estimated from reservoir size and the rate of carbon fluxes in and out.
In AP Environmental Science, you should be able to sort major carbon pools into short-term (fast-cycling) versus long-term (slow-cycling) storage and explain what makes them different.
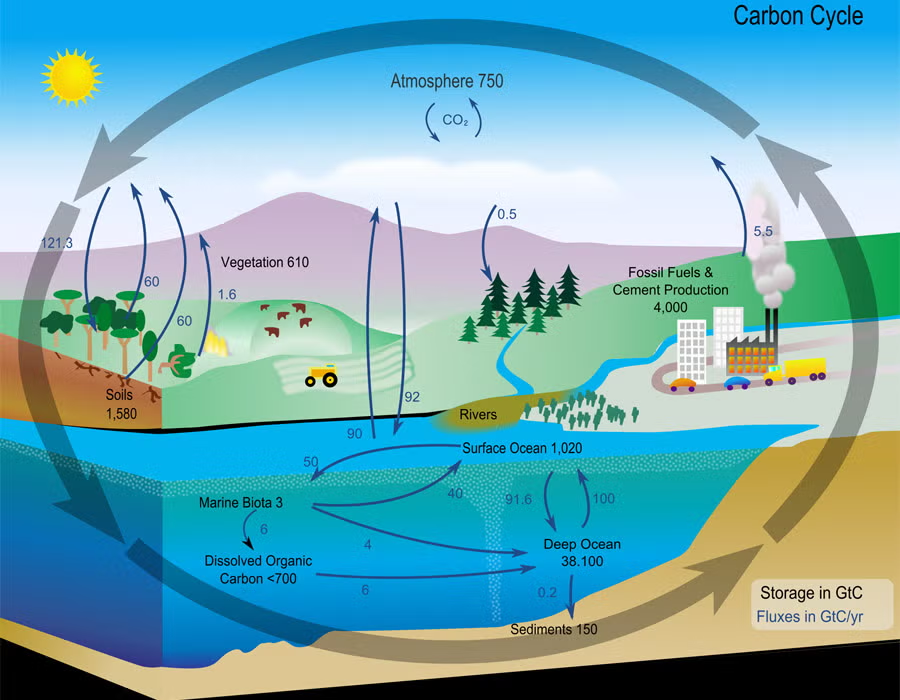
Quantitative schematic of Earth’s carbon reservoirs and the fluxes connecting them, with reservoir sizes (stored carbon) and transfer rates shown side-by-side. It helps distinguish fast-cycling near-surface pools (atmosphere, vegetation, surface ocean) from slower, larger pools (deep ocean, sediments, fossil carbon). The arrows reinforce that “storage time” (residence time) depends on exchange rates, not just how large the reservoir is. Source
Short-term (fast-cycling) carbon reservoirs
Short-term reservoirs store carbon for relatively brief periods (from days to decades, sometimes centuries) and exchange carbon readily with the atmosphere, water, and living organisms.
Key short-term reservoirs (typical examples)
Atmosphere: carbon mainly as (and small amounts as methane and other gases).
Living biomass: carbon in plants, algae, animals, and microbes as carbohydrates, lipids, and proteins.
Surface ocean and upper water layers: dissolved inorganic carbon (including in water and related forms).
Soil organic matter (active fraction): fresh litter and easily decomposed organic compounds.
Why storage times are short
Rapid biological turnover: organisms grow, respire, die, and decompose on short timescales.
Fast physical exchange: gases mix in the atmosphere; surface waters exchange gases with air.
High accessibility: carbon compounds are chemically available to decomposers and are not strongly locked into minerals.
What to notice about short-term reservoirs
They are sensitive to seasonal and annual changes (e.g., growing season uptake vs. winter release).
They respond quickly to disturbances because carbon is comparatively mobile and biologically reactive.
Long-term (slow-cycling) carbon reservoirs
Long-term reservoirs store carbon for thousands to millions of years, with much slower exchange rates.
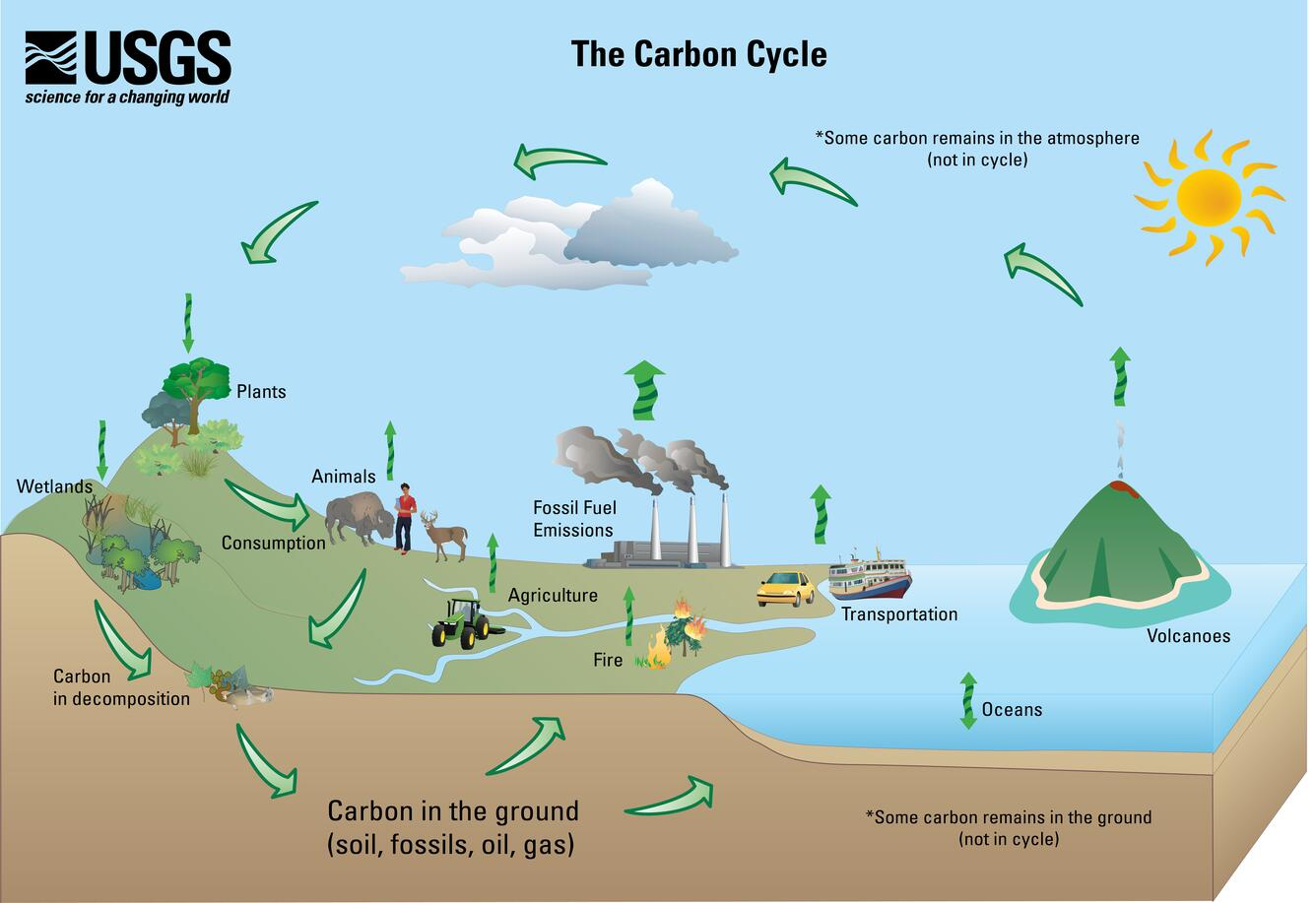
Conceptual diagram of the carbon cycle highlighting how carbon moves among atmosphere, biosphere, oceans, and geologic storage. The graphic is useful for linking long-term storage (carbon in the ground as soils/fossil deposits) to slow return pathways, while also showing rapid exchanges through living organisms and the surface environment. Human activities (fossil-fuel emissions and land use) are shown as direct transfers that move carbon from long-term storage into faster-cycling reservoirs. Source
Carbon here is typically physically isolated, chemically stable, or both.
Key long-term reservoirs (typical examples)
Sedimentary rocks (carbonate rocks): carbon stored as mineral carbonates (e.g., limestone).
Marine sediments and buried organic carbon: carbon trapped under accumulating sediments.
Fossil carbon deposits: coal, oil, and natural gas as concentrated forms of ancient organic carbon.
Deep ocean (often treated as longer-term relative to the atmosphere/surface ocean): large pool with slower mixing back to the surface.
Why storage times are long
Geologic sequestration: burial and rock formation isolate carbon from rapid cycling.
Slow conversion processes: transforming carbon into carbonate minerals or deep sediment layers takes long periods.
Limited exchange pathways: movement out of these reservoirs depends on slow processes such as uplift, weathering, or deep-ocean circulation.
What to notice about long-term reservoirs
They act as stabilising buffers in Earth’s carbon budget because the carbon is not easily exchanged.
Changes to these pools are generally slow, but when carbon does move out, it can shift other reservoirs substantially because the stored quantity is large.
Comparing short-term vs long-term storage (what AP expects)
Quick comparison anchors
Timescale:
Short-term: days to decades (sometimes centuries)
Long-term: thousands to millions of years
Dominant controls:
Short-term: biology + near-surface physics
Long-term: geology + deep isolation
Carbon form:
Short-term: organic matter and dissolved/atmospheric
Long-term: carbonates and buried reduced carbon
Mobility:
Short-term: high (frequent fluxes)
Long-term: low (restricted fluxes)
A useful way to think about it
A reservoir can be large but still “slow” if its carbon is difficult to access and exchange (for example, carbonate rock). A reservoir can be smaller but “fast” if carbon is constantly entering and leaving (for example, living biomass).
FAQ
They combine reservoir size estimates with measured or modelled fluxes in and out.
Methods include atmospheric monitoring, ocean tracer studies, and isotopic approaches such as $^{13}C$ and $^{14}C$.
Different soil carbon fractions cycle at different speeds.
Fast pool: fresh litter and microbial biomass
Slow pool: carbon protected inside aggregates or bound to minerals
Deep waters mix back to the surface slowly.
Isolation from the atmosphere and slow thermohaline circulation increase storage time even though the carbon remains dissolved rather than mineralised.
Carbonate rock stores carbon as mineral carbonate ions in solid lattices, making it chemically stable.
Organic deposits store carbon in reduced organic molecules, which are stable mainly because they are buried and oxygen-limited.
Cold, waterlogged conditions slow decomposition, allowing carbon to accumulate for long periods.
Stability depends on ground temperature, ice content, drainage, and microbial activity once thaw begins.
Practice Questions
State one example of a short-term carbon reservoir and one example of a long-term carbon reservoir. (2 marks)
1 mark for a correct short-term reservoir (e.g., atmosphere, living biomass, surface ocean, active soil organic matter).
1 mark for a correct long-term reservoir (e.g., carbonate rocks, marine sediments, fossil carbon deposits, deep ocean).
Compare short-term and long-term carbon reservoirs in terms of (i) typical storage time, (ii) the main processes controlling carbon exchange, and (iii) the chemical form in which carbon is commonly stored. (5 marks)
1 mark: correct comparison of storage time (short: days–decades; long: thousands–millions of years).
1 mark: short-term exchange controlled mainly by biological activity and rapid atmosphere–surface water interactions.
1 mark: long-term exchange controlled mainly by geological burial/rock formation and slow physical circulation.
1 mark: carbon form in short-term reservoirs commonly as organic matter and/or atmospheric/surface-water .
1 mark: carbon form in long-term reservoirs commonly as carbonates and/or buried reduced carbon (fossil carbon, sediments).

