AP Syllabus focus:
‘Explain how decomposition stored carbon over millions of years and how burning fossil fuels rapidly transfers that stored carbon to the atmosphere as carbon dioxide.’
Decomposition links the short-term carbon in dead biomass to long-term carbon storage in soils, sediments, and eventually fossil fuels.
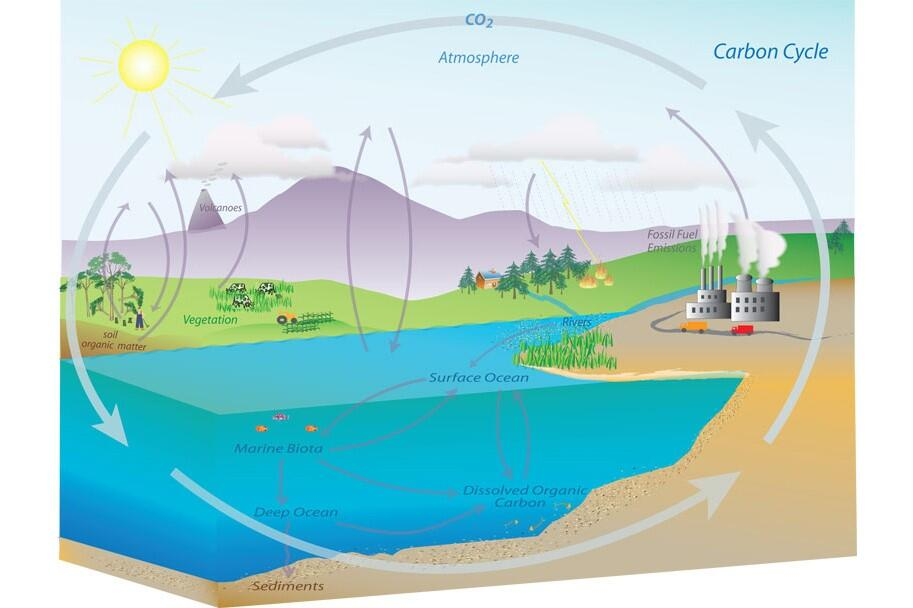
This carbon-cycle diagram summarizes the major carbon reservoirs (atmosphere, biosphere, ocean, and geologic stores) and the fluxes that move carbon among them. It helps distinguish rapid biological processes (including decomposition and respiration) from slower transfers into long-term storage such as sediments and fossil carbon, supporting the AP focus on mismatched timescales. Source
Human extraction and combustion short-circuit this slow pathway, increasing atmospheric CO₂ rapidly.
Decomposition and Carbon Storage
What decomposition does to carbon
When organisms die or produce waste, decomposers (mainly bacteria and fungi) break complex organic molecules into simpler substances. During this process, carbon follows two main paths: it is returned to the atmosphere as CO₂, or it is retained and stabilized in soils or sediments.
Decomposition: The biological breakdown of dead organic matter and wastes into simpler compounds by microbes and detritivores, transferring carbon into CO₂ and/or longer-lived soil and sediment carbon pools.
Even though decomposition often releases CO₂, it can also contribute to long-term storage when breakdown is incomplete or when carbon becomes physically or chemically protected.
Why some carbon persists (slow carbon pathways)
Long-term storage is favored when conditions reduce microbial activity or limit oxygen, slowing decay and allowing carbon-rich material to accumulate.
Key controls that promote carbon retention:
Low oxygen (anoxic) conditions: waterlogged soils, wetlands, lake bottoms, ocean sediments
Low temperatures: slow microbial metabolism in cold environments
Rapid burial: sediments cover organic material, isolating it from oxygen
Recalcitrant compounds: some plant materials resist decay (e.g., lignin-rich tissues)
Soil protection: organic carbon binds to minerals or becomes trapped in soil aggregates
Over long timescales, these mechanisms shift carbon from fast-cycling biomass into sedimentary organic carbon, lowering the fraction that returns quickly to the atmosphere.
From partially decomposed matter to peat and organic-rich sediments
In persistently wet, low-oxygen environments, plant material can accumulate as peat or other organic-rich deposits because decomposition cannot keep pace with litter input. If burial continues, heat and pressure gradually transform this stored carbon into more stable forms.
Fossil Fuels: Carbon Stored Over Millions of Years
Formation of fossil fuels (geologic carbon storage)
Fossil fuels form when large quantities of organic matter are buried and altered over millions of years.
This staged diagram shows how organic matter can accumulate, be buried by sediments, and eventually be transformed into fossil fuels under heat and pressure over very long timescales. It reinforces the idea that fossil fuel carbon represents ancient organic carbon that escaped complete decomposition and was sequestered in geologic reservoirs. Source
Decomposition is central because it determines how much organic carbon escapes complete breakdown and becomes available for long-term sequestration.
Fossil fuels: Energy-rich carbon compounds (coal, petroleum, natural gas) formed from buried organic matter transformed by heat and pressure over geologic time, storing carbon in long-term reservoirs.
This slow sequence depends on limited oxygen, sustained burial, and continued sediment deposition, which together prevent most carbon from being returned to the atmosphere on ecological timescales.
Typical origins (high-level)
Coal: mainly from terrestrial plant material accumulating in swampy settings, often beginning as peat
Petroleum and natural gas: largely from aquatic microorganisms and plankton buried in marine or lake sediments
The crucial AP idea is the timescale: carbon locked into fossil fuels represents ancient atmospheric carbon that avoided full decomposition and was stored far longer than living biomass or most soil organic matter.
Burning Fossil Fuels and Atmospheric CO₂
Rapid transfer from long-term storage to the atmosphere
Combustion oxidizes reduced carbon in fossil fuels, producing CO₂ (and water), releasing energy, and moving carbon from a long-term geologic reservoir into the atmosphere in years to centuries—far faster than the millions of years required for formation.
Major features of this transfer:
Direction: fossil fuel reservoir → atmosphere (as CO₂)
Speed: rapid compared with geologic sequestration
Scale: large, cumulative emissions from electricity generation, transportation, industry, and heating
Chemical outcome: more atmospheric CO₂ available to participate in climate-relevant processes
Why the rate matters (mismatch of timescales)
Natural burial and fossil fuel formation remove carbon extremely slowly, while modern combustion releases it quickly. This creates a net atmospheric increase because:
the input rate of CO₂ from burning fossil fuels greatly exceeds the long-term removal rate by sediment burial and fossil formation
ecosystems and oceans can absorb only part of the added CO₂ on short timescales, leaving an atmospheric surplus
Decomposition vs fossil fuel combustion: same element, different tempo
Both processes move carbon and can produce CO₂, but they differ in the reservoir being tapped:
Decomposition: mainly cycles recent carbon in biomass and soils
Fossil fuel burning: mobilizes ancient carbon that was removed from active cycling for geologic time
Understanding this difference explains why fossil fuel combustion is a powerful driver of rising atmospheric CO₂ despite decomposition being a natural, ongoing source of CO₂.
FAQ
Drainage adds oxygen, accelerating microbial breakdown.
Faster decomposition converts stored peat carbon into $CO_2$ (and sometimes $CH_4$ during transitions).
Physical disturbance also exposes deeper carbon to decay.
Not all persistence is due to anoxia.
Organic matter can bind to clay and metal oxides.
Soil aggregates can physically shield carbon from microbes and enzymes.
The key factor is oxygen availability.
With oxygen: microbes produce mostly $CO_2$.
Without oxygen: anaerobic pathways can generate more $CH_4$.
No; carbon intensity varies.
Coal generally emits more $CO_2$ per unit energy than oil.
Natural gas is often lower, though methane leakage can offset benefits.
CCS aims to interrupt the transfer to the atmosphere.
It captures $CO_2$ from flue gases and injects it into geologic formations.
Effectiveness depends on capture rates, storage integrity, and long-term monitoring.
Practice Questions
Describe how decomposition can lead to long-term carbon storage that may eventually form fossil fuels. (2 marks)
Mentions incomplete decomposition under low-oxygen/cold/waterlogged conditions leading to accumulation/burial of organic matter (1)
Links continued burial/heat/pressure over millions of years to formation of fossil fuels (1)
Explain how burning fossil fuels rapidly transfers stored carbon to the atmosphere as carbon dioxide, and why this increases atmospheric compared with natural long-term storage processes. (6 marks)
Identifies fossil fuels as long-term (geologic) carbon stores formed over millions of years (1)
States combustion oxidises fossil carbon, producing (1)
Explains that combustion releases carbon on short human timescales (years–centuries) (1)
Contrasts with slow natural sequestration via burial/sedimentation/fossil formation (1)
Explains the rate imbalance: emissions exceed long-term removal, so atmospheric accumulates (1)
Clear linkage between reservoir transfer (geologic → atmosphere) and net atmospheric increase (1)

