AP Syllabus focus:
‘Describe how many nitrogen reservoirs hold nitrogen compounds for relatively short periods of time.’
Nitrogen moves through several reservoirs, but unlike carbon in fossil fuels, much of Earth’s usable nitrogen is stored briefly. Understanding typical storage times helps explain rapid ecosystem responses to disturbance.
Core idea: reservoirs and storage time
What “storage time” means
Nitrogen can be stored in multiple reservoirs (locations where nitrogen-containing matter accumulates), but many have short residence times because nitrogen is biologically reactive and frequently transformed.
Reservoir: A place in the environment where a substance (such as nitrogen compounds) is stored (e.g., atmosphere, soil, biomass, oceans).
Storage time depends on:
Chemical form (e.g., nitrate vs organic nitrogen)
Biological demand (plant uptake, microbial metabolism)
Environmental conditions (oxygen, moisture, temperature)
Disturbance and human inputs (fertiliser, land-use change)
A useful contrast is fast-cycling reservoirs (days to decades) versus slow-cycling reservoirs (centuries or longer). This subtopic emphasises that many nitrogen reservoirs are fast-cycling.
Residence time (typical storage time)
Because nitrogen is continually transformed (especially by microbes), a given nitrogen atom often does not remain long in the same reservoir.
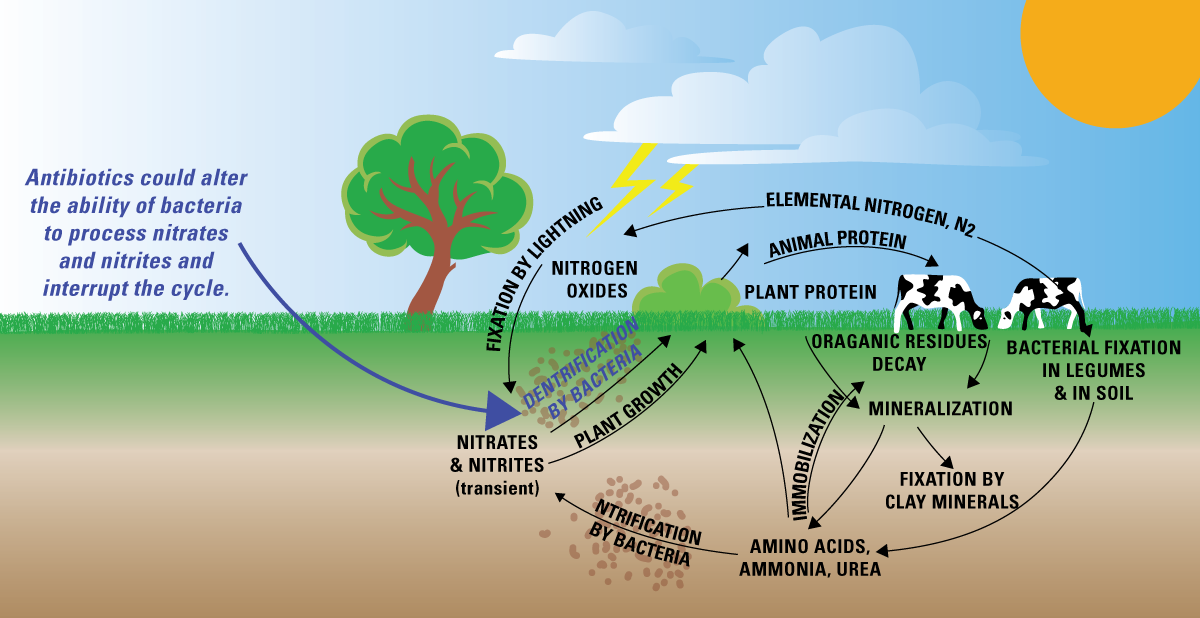
This diagram summarizes the terrestrial nitrogen cycle by showing nitrogen’s major biological reservoirs (plant and animal protein, organic residues, and soil nitrogen pools) and the microbially mediated processes that move nitrogen among them. It highlights rapid conversions among reactive forms (e.g., nitrification and denitrification) that help explain short residence times in many ecosystem reservoirs. Source
Residence time (storage time): The average time a substance remains in a reservoir before leaving for another reservoir.
Major nitrogen reservoirs with typical storage times
Living biomass (plants, animals, microbes): short storage
Nitrogen in living tissue (proteins, DNA, chlorophyll) is usually stored for:
Days to years in microbes and annual plants (rapid turnover)
Years to decades in long-lived plants (wood, perennial tissues)
Generally short because death, predation, and waste quickly move nitrogen to detritus and soils
Key implication: Ecosystem nitrogen availability can change quickly when productivity or species composition changes.
Detritus and soil organic matter: short to moderate storage
A large amount of nitrogen is stored in dead organic matter and soil organic nitrogen.
Weeks to years in fresh litter and detritus (fast decomposition under warm, moist conditions)
Decades to centuries in more stable soil organic matter (protected within soil aggregates or bound to minerals)
Even when soil storage is longer than biomass, it is still often considered relatively short on geologic timescales and can be rapidly altered by cultivation, erosion, and drainage.
Dissolved inorganic nitrogen in soils and waters: very short storage
Forms such as ammonium () and nitrate () commonly have short storage times:
Often days to months in soils because they are quickly:
taken up by plants and microbes
transformed by microbes
leached to groundwater or runoff to surface waters (especially nitrate)
This short storage helps explain why fertiliser nitrogen can quickly appear downstream and why nitrogen availability can fluctuate strongly within a growing season.
Atmosphere (as ): large reservoir, but “reactive” nitrogen is brief
While atmospheric is abundant, most organisms cannot use it directly, and the focus for “short periods” is often on reactive nitrogen (e.g., , aerosols).
Reactive nitrogen in the air typically persists hours to days before deposition (rainout/dry deposition)
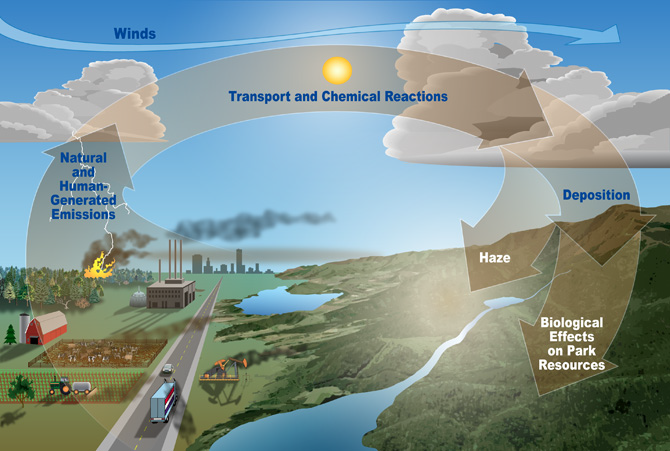
This diagram illustrates the pathway from nitrogen emissions to atmospheric transport and chemical reactions, followed by wet and dry deposition onto land and water. It reinforces why many atmospheric reactive nitrogen species (e.g., -derived products) have short atmospheric residence times: they are efficiently removed by deposition processes. Source
Deposited nitrogen then enters fast-cycling terrestrial or aquatic reservoirs
Oceans and aquatic systems: variable storage
Nitrogen in aquatic systems can be:
Short in surface waters (days to years) due to uptake by phytoplankton and rapid recycling
Longer in deep waters (decades to centuries) due to slower mixing, but still commonly shorter than geologic storage
In many watersheds, nitrogen can be quickly transported away, so local storage is often limited unless conditions favour accumulation.
Sediments and rocks: longer-term nitrogen storage (but less “active”)
Some nitrogen becomes buried in sediments as organic nitrogen or fixed in minerals.
Centuries to millions of years (slow cycling)
This reservoir is typically less involved in day-to-day ecosystem nitrogen availability compared with biomass, soils, and dissolved forms
Why “many reservoirs” are short-term in practice
Fast turnover is driven by:
Microbial transformations that rapidly convert nitrogen among forms
High biological demand for nitrogen in growth
Mobility of nitrate in water leading to quick transport and loss
Disturbance sensitivity: soil disturbance and added nitrogen inputs can shift storage from soil/biota into waterways and the atmosphere quickly
FAQ
Warm, moist conditions generally speed microbial activity and decomposition, shortening storage in litter and soil organic pools.
Cold or dry conditions slow decomposition, allowing nitrogen to remain longer in organic matter.
Nitrate ($NO_3^-$) is negatively charged and is less likely to bind to soil particles, so it moves with water.
Ammonium ($NH_4^+$) is positively charged and can be held on cation-exchange sites, increasing retention.
Stability increases when organic matter is:
physically protected inside soil aggregates
chemically complex (harder for microbes to break down)
bound to mineral surfaces (reduced accessibility)
Clearing vegetation can reduce plant uptake and increase erosion, shifting nitrogen from biomass/soil into runoff.
Tilling can aerate soil and accelerate decomposition, moving nitrogen out of long-lived organic pools into fast-cycling forms.
Nitrogen can be quickly recycled by algae and microbes, but also exported by outflow or converted to gaseous forms under low-oxygen conditions.
This combination can limit how long reactive nitrogen remains in the water column.
Practice Questions
State two nitrogen reservoirs that typically store nitrogen compounds for relatively short periods of time. (2 marks)
Any two valid reservoirs with short storage, e.g. living biomass (1), soil organic matter/detritus (1), dissolved inorganic nitrogen in soils/waters (1), reactive nitrogen in the atmosphere (1).
Explain why nitrogen stored as nitrate in soils often has a shorter storage time than nitrogen stored in stable soil organic matter. (5 marks)
Nitrate is highly soluble/mobile in water, so it can be leached/run off (1)
Nitrate is readily taken up by plants/microbes (1)
Nitrate is rapidly transformed by microbes (e.g. converted to other forms) (1)
Stable soil organic matter is physically/chemically protected (e.g. within aggregates or bound to minerals) (1)
Stable organic matter decomposes more slowly, extending storage time (1)

