AP Syllabus focus:
‘Define the nitrogen cycle as the movement of nitrogen-containing atoms and molecules between sources and sinks.’
Nitrogen is essential for proteins and DNA, but most organisms cannot use atmospheric nitrogen directly. The nitrogen cycle tracks nitrogen’s movement among reservoirs, highlighting where it is produced, stored, and removed.
What the nitrogen cycle describes
Nitrogen cycle: The movement of nitrogen-containing atoms and molecules between sources and sinks in Earth systems.
In AP Environmental Science, the nitrogen cycle is about following nitrogen as it changes chemical form and shifts location among the atmosphere, living things, soils, and waters.
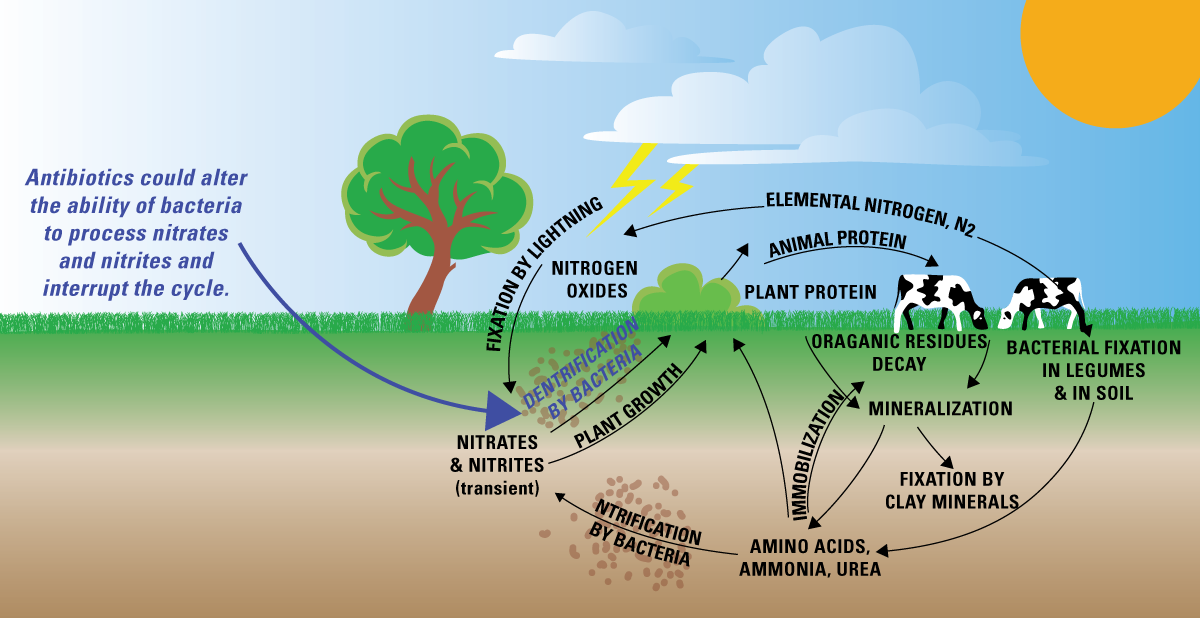
This labeled diagram summarizes the major nitrogen reservoirs and the transformations that connect them, including fixation of atmospheric , conversion among inorganic forms (nitrification), and return of nitrogen to the atmosphere (denitrification). It reinforces that “movement” in the nitrogen cycle includes both transport between reservoirs and chemical changes driven largely by microbes. Source
Key language: sources vs sinks
Source: A process or place that releases a substance (here, nitrogen compounds) into a system faster than it removes it.
A nitrogen source increases the amount of nitrogen in a particular reservoir (for example, adding nitrate to a stream).
Sink: A process or place that removes a substance from a system faster than it adds it.
A sink decreases the amount of nitrogen in a reservoir (for example, removing dissolved nitrate from water into sediments or biomass).
Major chemical forms of nitrogen (why “movement” is more than transport)
Nitrogen “moves” both by physical transport (wind, water flow) and by chemical change.
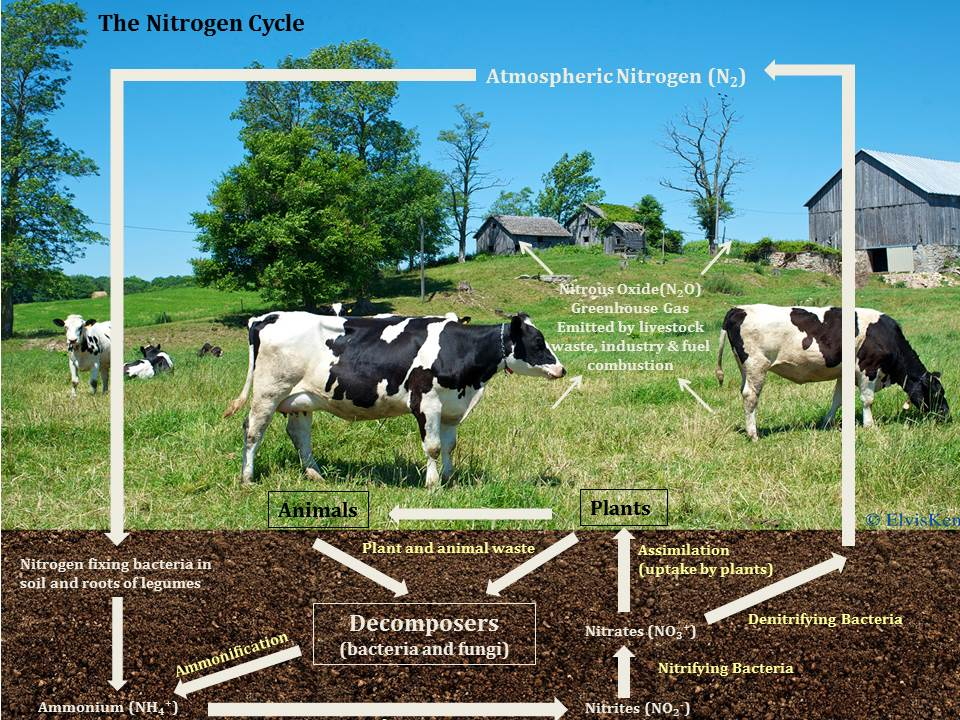
This diagram emphasizes the step-by-step conversions among nitrogen forms in ecosystems, highlighting microbial processes (ammonification, nitrification, and denitrification) and plant uptake (assimilation). The labeled arrows connect soil nitrogen pools (e.g., and ) to biomass and back to atmospheric , making the chemistry of the cycle visually explicit. Source
Key forms students should recognise include:
(nitrogen gas): abundant in air, relatively unusable to most organisms without microbial conversion
Organic nitrogen: nitrogen bound in biomass (proteins, nucleic acids) and detritus
Ammonium () and ammonia (): reduced forms that can be held on soils and taken up by some producers
Nitrate (): oxidised, highly soluble form that moves readily with water (and is therefore easy to lose from soils)
Reactive nitrogen: Nitrogen in biologically and chemically available forms (such as and ) that can readily cycle through ecosystems.
Reactive nitrogen is the part of the nitrogen cycle most directly linked to plant growth, water quality, and ecosystem change.
Nitrogen sources and sinks: where nitrogen enters and leaves reservoirs
Natural sources (add nitrogen to a reservoir)
Natural sources are processes that increase nitrogen availability in a given place:
Microbial conversions that make nitrogen available to producers (creating more reactive nitrogen in soils and waters)
Decomposition of dead organisms and waste, which returns organic nitrogen to soils and sediments as simpler nitrogen compounds
Atmospheric deposition from natural events (for example, lightning can create nitrogen oxides that later enter ecosystems via precipitation)
Natural sinks (remove nitrogen from a reservoir)
Natural sinks reduce nitrogen within a specific reservoir or reduce reactive nitrogen back to less available forms:
Plant and microbial uptake (assimilation): moves inorganic nitrogen (often or ) into biomass, shifting nitrogen from soil/water into living tissue
Burial and sediment storage: nitrogen becomes part of longer-lived organic matter in sediments, reducing short-term availability in water
Microbial return to atmospheric forms: certain microbial pathways convert reactive nitrogen back toward gaseous forms, removing it from soils/waters into the atmosphere
Human influences on nitrogen sources and sinks (conceptual, source–sink framing)
Human activities often act as additional sources of reactive nitrogen in ecosystems:
Synthetic fertilisers add large amounts of plant-available nitrogen to soils
Combustion (vehicles, power plants) emits nitrogen oxides that can be deposited downwind
Wastewater and manure inputs add nitrogen to waterways and coastal zones
Humans can also strengthen or weaken sinks:
Harvesting crops can remove nitrogen stored in biomass from fields (a sink from the farm ecosystem’s perspective)
Wetlands can act as important sinks by removing dissolved nitrogen from moving water, depending on conditions
Soil disturbance may reduce the ability of soils to retain nitrogen, weakening sink strength and increasing losses
Why sources and sinks matter for environmental science
Thinking in sources and sinks helps predict:
Nutrient availability and productivity: adding sources of reactive nitrogen can increase plant growth, but may shift species composition
Transport to water bodies: when sources exceed sinks in soils, soluble forms (especially ) are more likely to move into groundwater, streams, and lakes
Ecosystem balance: changing sink capacity (for example, removing wetlands) alters where nitrogen accumulates and where it is exported
FAQ
Soils with high clay content and organic matter tend to hold more $NH_4^+$ and organic nitrogen, strengthening sink behaviour.
Sandy or compacted soils often allow faster water movement, increasing export of dissolved $NO_3^-$ and making the system behave more like a source to nearby waters.
$NO_3^-$ is negatively charged and is less strongly held by many soil particles, so it travels easily with percolating water.
$NH_4^+$ is positively charged and is more readily retained on exchange sites, so it is often less mobile.
Deposition is the transfer of nitrogen compounds from air to land/water via rain, snow, fog, or dry particles.
It tends to be higher near urban/industrial areas and downwind of major emission sources because nitrogen oxides and related compounds are produced during combustion.
Water level, temperature, and oxygen availability shift through the year, changing which microbial processes dominate.
Seasonal plant growth also matters: rapid growth increases assimilation into biomass, while dieback can later release nitrogen back into sediments and water.
They often use mass balance approaches comparing nitrogen inputs (fertiliser sales, deposition estimates, upstream loads) with outputs (river export, harvest removal).
Isotopic tracers (e.g., $\delta^{15}N$ patterns) can help distinguish among nitrogen sources such as sewage, manure, and synthetic fertiliser.
Practice Questions
Define the nitrogen cycle in terms of sources and sinks. (2 marks)
States that it is the movement of nitrogen-containing atoms/molecules (1)
Explicitly links movement to sources and sinks (release vs removal) (1)
Using the ideas of sources and sinks, explain how human activity can increase reactive nitrogen in a river system and describe two possible sink pathways that could reduce it. (6 marks)
Identifies at least one human source adding nitrogen to the system (e.g., fertiliser runoff, wastewater, atmospheric deposition from combustion) (1)
Explains that a source increases nitrogen in the river or its catchment faster than removal (1)
Correctly names reactive nitrogen forms or examples (e.g., , ) (1)
Describes sink pathway 1 with correct direction of movement/removal (e.g., uptake into biomass; burial in sediments; removal in wetlands) (1)
Describes sink pathway 2 with correct direction of movement/removal (1)
Links sink action to reduced concentration/availability in the river water (1)

