AP Syllabus focus:
‘Identify the atmosphere as the major reservoir of nitrogen and explain why it dominates the global nitrogen pool.’
Nitrogen is essential to proteins and DNA, yet most of Earth’s nitrogen is not immediately usable by organisms. Understanding the atmosphere’s role as the main reservoir explains nitrogen scarcity in ecosystems.
The atmosphere as Earth’s dominant nitrogen store
What “main reservoir” means in the nitrogen cycle
Reservoir: a major storage location for a substance (here, nitrogen) where it can be held for variable lengths of time before moving to other parts of the system.
In the nitrogen cycle, the atmosphere is the largest reservoir because it contains an overwhelming share of Earth’s nitrogen in a stable, long-lasting form.
Why atmospheric nitrogen is so abundant
The atmosphere is about 78% nitrogen gas () by volume.
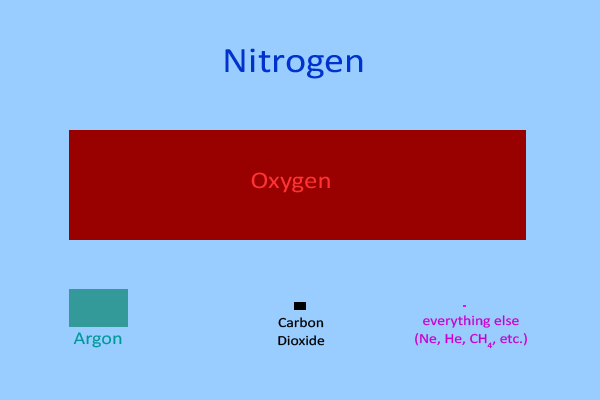
This diagram summarizes the relative abundances of gases in Earth’s (dry) atmosphere, highlighting nitrogen as the dominant component. It supports the idea that the atmosphere is an enormous, well-mixed reservoir because most atmospheric molecules are . Source
This makes it the dominant global nitrogen pool for three related reasons:
Sheer size and mixing
The atmosphere has a very large total mass and is well mixed by global circulation.
Because is widespread and continuously mixed, it forms a single, planet-scale store rather than many small, isolated stores.
Chemical stability of
Atmospheric nitrogen is mainly diatomic nitrogen (), which is highly unreactive due to its strong bond.
This stability means nitrogen remains in the atmosphere for long periods unless converted by specialised processes, so the reservoir persists and accumulates.
Slow biological access
Most organisms cannot use directly; nitrogen must be converted into biologically available forms.
Because relatively few pathways move nitrogen out of the atmosphere at any given time, the atmospheric pool stays large compared with living biomass, soils, or waters.
Why the atmospheric reservoir dominates the global nitrogen pool
Limited pathways remove from air
Only certain processes transfer nitrogen from the atmospheric reservoir into other reservoirs at meaningful rates:
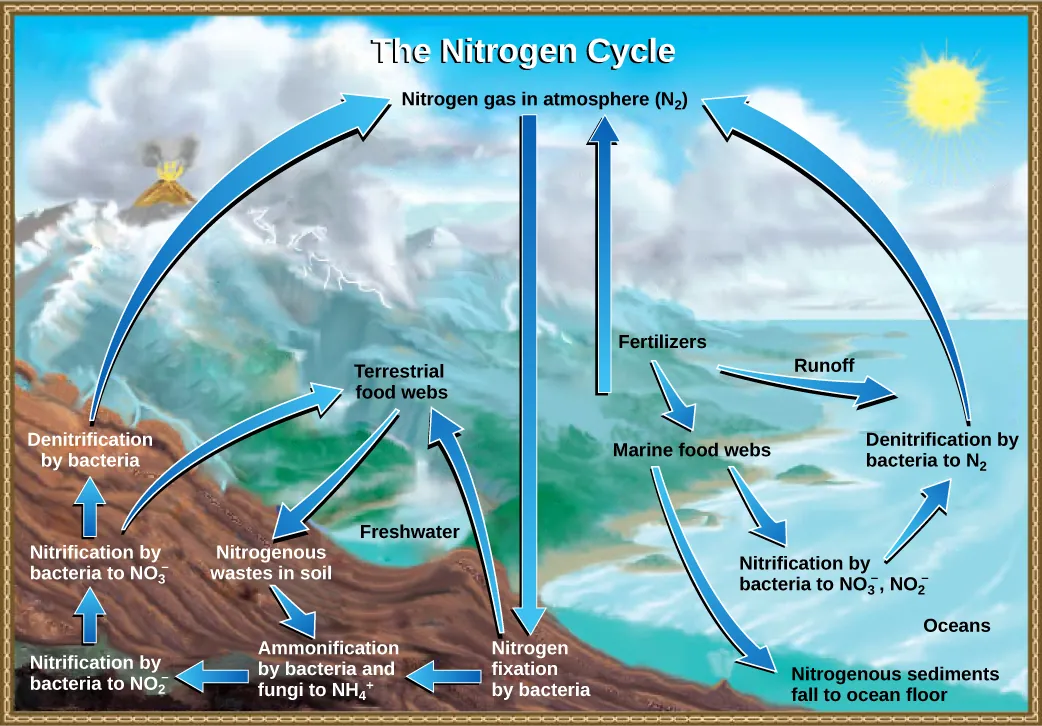
This nitrogen cycle diagram traces how atmospheric enters ecosystems primarily through nitrogen fixation, then moves through ammonification and nitrification, and returns to the atmosphere via denitrification. Seeing the full loop makes it clear why the atmospheric reservoir remains dominant: most nitrogen ultimately cycles back to in the air. Source
Biological nitrogen fixation
Carried out by certain microbes (including symbiotic bacteria associated with some plants).
Converts atmospheric into reduced nitrogen compounds that can enter food webs.
Abiotic fixation
Lightning provides energy to form reactive nitrogen compounds that can be deposited to land and water.
This pathway is globally important but still small relative to the total atmospheric supply.
Industrial fixation
Human activity can fix nitrogen for fertilizers, but even large human flows do not reduce the atmospheric fraction much because the atmospheric reservoir is so vast.
Because these removal pathways are limited compared with the size of the atmospheric store, the atmosphere remains the major reservoir even while nitrogen continuously cycles through ecosystems.
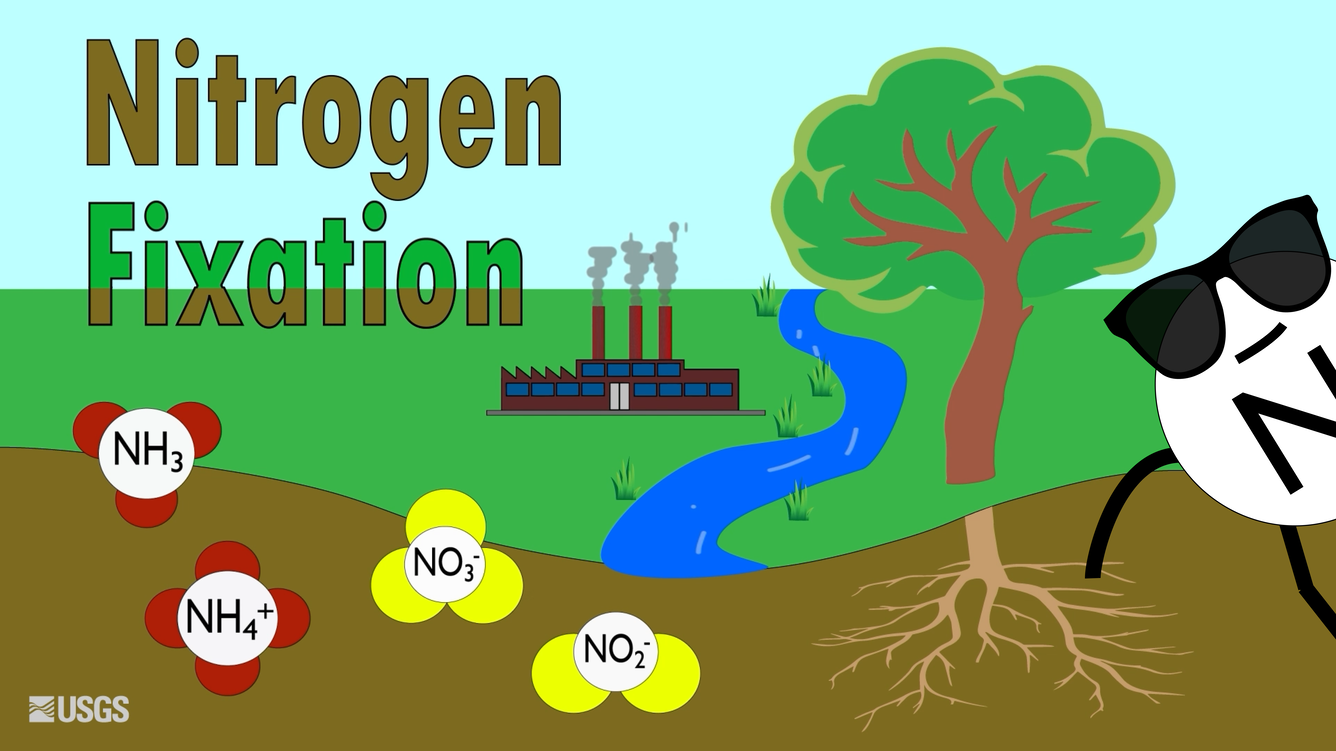
This USGS illustration contrasts natural and industrial nitrogen fixation pathways that move nitrogen out of atmospheric and into reactive forms. It highlights that these conversion pathways are specialized and rate-limited compared with the sheer size of the atmospheric pool, which helps explain why the atmosphere dominates the global nitrogen reservoir. Source
Other reservoirs are comparatively small and more dynamic
Although nitrogen is found in soils, organisms, and aquatic systems, these stores generally:
contain far less nitrogen than the atmosphere overall
change more quickly due to biological uptake, decomposition, and water movement
can be locally important for productivity, even if globally minor compared with atmospheric
This contrast is central to why the atmosphere dominates: most nitrogen is “parked” in a chemically stable atmospheric form, while the biologically active fraction circulates through smaller, faster-turnover reservoirs.
Implications for ecosystems and environmental management
Atmospheric dominance does not mean nitrogen is always available
Because is largely unusable to most life, ecosystems may experience nitrogen limitation even though nitrogen is abundant in the air. Key implications include:
Ecosystem productivity depends on conversion capacity
Local availability often hinges on the presence and activity of nitrogen-fixing microbes and the conditions that support them.
Sensitivity to changes in fixation and deposition
Shifts in fixation rates or atmospheric deposition can strongly affect ecosystem nutrient balance because the usable nitrogen pool is relatively small compared with the atmospheric store.
Separation between “amount” and “access”
The atmosphere dominates the global nitrogen pool by quantity, but ecological outcomes depend on how effectively nitrogen moves out of and into biologically available forms.
FAQ
They combine atmospheric composition measurements (fraction of $N_2$) with the total mass of the atmosphere.
This yields a global nitrogen mass estimate that is far larger than nitrogen stored in soils, oceans, or biomass.
$N_2$ reacts when enough energy is available to break its strong bond.
High-energy events: lightning
Enzyme-mediated reactions: microbial nitrogenase (biological fixation)
It varies very little over short timescales because the reservoir is enormous.
Even substantial human fixation mostly redistributes nitrogen among non-atmospheric pools rather than noticeably shrinking atmospheric $N_2$.
Air circulates globally, mixing gases across hemispheres and continents.
Soils are fragmented into local patches with different chemistry and biology, creating many smaller, less connected nitrogen stores.
Yes. Pollution can increase reactive nitrogen species (collectively “$N_r$”) such as nitrogen oxides.
These changes can strongly affect deposition and ecosystems, while total atmospheric $N_2$ remains essentially unchanged.
Practice Questions
State the main reservoir of nitrogen in the nitrogen cycle and name the predominant chemical form found there. (2 marks)
Atmosphere/air is the main reservoir (1)
Nitrogen gas, (1)
Explain why the atmosphere dominates the global nitrogen pool, despite nitrogen being essential to living organisms. (5 marks)
Most atmospheric nitrogen is present as and the atmosphere is ~78% nitrogen (1)
is chemically stable/inert, so it persists and accumulates in the air (1)
Most organisms cannot use directly; it must be fixed into usable forms (1)
Only limited processes remove from the atmosphere (e.g., nitrogen-fixing microbes, lightning, industrial fixation) (1)
Other reservoirs (biomass/soils/waters) are much smaller and/or have faster turnover, so they do not rival the atmospheric store (1)

