AP Syllabus focus:
‘Explain why the phosphorus cycle lacks an atmospheric component, making phosphorus naturally scarce; in undisturbed ecosystems it is often the limiting nutrient in biological systems.’
Phosphorus is essential for DNA, ATP, and cell membranes, yet ecosystems often have too little available phosphorus. Its unique chemistry keeps it out of the atmosphere, slowing recycling and frequently limiting biological growth.
Why the Phosphorus Cycle Has No Atmospheric Phase
Phosphorus moves mainly as phosphate ions in water, soil, rocks, and living tissue. Unlike carbon or nitrogen, phosphorus does not readily form common gaseous compounds under Earth-surface conditions.
Chemical and physical reasons
Low volatility: Most phosphorus occurs bound in solid minerals (e.g., apatite) or dissolved as phosphate; these forms do not evaporate into air.
Strong binding to particles: Phosphate tends to adsorb to soil and sediment surfaces, so it is transported primarily by erosion and runoff rather than by wind as a gas.
No large gaseous reservoir: Because there is effectively no stable atmospheric pool, there is no rapid, global “mixing” step that can quickly redistribute phosphorus.
What “no atmospheric phase” means for cycling speed
Without an atmospheric pathway, most phosphorus cycling depends on comparatively slow processes:
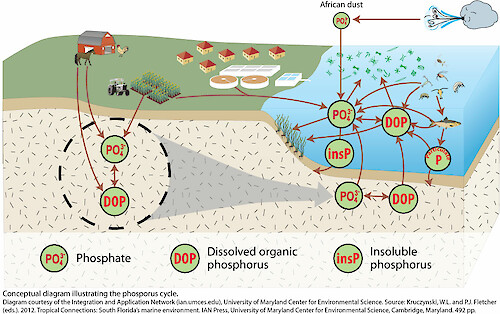
Conceptual diagram of the phosphorus cycle showing phosphorus pools (e.g., phosphate, dissolved organic phosphorus, and insoluble phosphorus) and transfers among land, freshwater, and marine environments. The figure visually reinforces that phosphorus primarily moves via weathering, runoff, biological uptake, and sedimentation rather than through an atmospheric gas phase. Source
Weathering of rocks releases phosphate into soils and surface waters.
Uptake by producers incorporates phosphorus into biomass.
Decomposition returns phosphorus to soils and water.
Sedimentation and burial lock phosphorus into sediments and rocks for long periods.
This makes phosphorus supply strongly dependent on local geology and soil development, rather than fast planet-wide exchange.
Phosphorus Scarcity in Ecosystems
Because phosphorus is often trapped in insoluble minerals or buried in sediments, the amount that is biologically available at any time can be small.
Why “naturally scarce” is a common outcome
Slow inputs: New phosphorus enters ecosystems mainly through rock weathering, which is slow compared with biological demand.
Losses are hard to replace: Phosphorus can be washed away in runoff attached to sediments, and there is no atmospheric reservoir to “refill” the system quickly.
Soil age matters: Over long periods, soils can become depleted as phosphorus is leached or exported, especially where replenishment from fresh mineral material is limited.
Phosphorus as the Limiting Nutrient (Undisturbed Ecosystems)
In many undisturbed ecosystems, phosphorus is the nutrient most likely to run out first relative to organism needs, constraining growth even when other requirements (light, water, nitrogen) are sufficient.
Limiting nutrient: The essential nutrient in shortest supply relative to demand that restricts growth, reproduction, or primary productivity in a biological system.
A key implication is that small changes in phosphorus availability can cause noticeable changes in primary productivity and community structure because organisms cannot substitute another element for phosphorus in critical molecules (ATP, nucleic acids, phospholipids).

Aerial photograph of the Experimental Lakes Area Lake 226 split-basin experiment, where phosphorus was added to one side of the lake, triggering a dramatic algal bloom. The side without phosphorus addition stayed comparatively clear, illustrating how phosphorus can be the limiting nutrient controlling primary productivity in aquatic ecosystems. Source
How limitation shapes biological systems
Producer growth constraints: Plants and algae may grow slowly or remain low-biomass when phosphorus is scarce.
Competitive outcomes: Species better at acquiring or conserving phosphorus can be favored.
Tight recycling: In phosphorus-limited systems, decomposition and uptake often form a closely coupled loop, because organisms rapidly reuse any available phosphate.
“Often” does not mean “always”
Phosphorus limitation is common in undisturbed settings because of the absence of atmospheric replenishment, but which nutrient limits growth can vary with local conditions (e.g., soil mineral content and how easily phosphate is released).
FAQ
Yes, but mostly as dust or soil particles containing phosphate, not as a gas.
Long-distance dust transport occurs, yet it is generally smaller and less consistent than atmospheric cycling of gaseous elements.
Phosphate can become less available when it binds tightly to mineral surfaces.
Acidic soils: stronger binding to iron/aluminium oxides
Alkaline soils: precipitation with calcium compounds
Over long timescales, easily weathered minerals are depleted and phosphorus is gradually exported or locked into stable forms.
With little fresh mineral input, replenishment remains slow.
Internal loading is the release of phosphate from bottom sediments back into the water column under certain conditions (often low oxygen).
It can sustain phosphate availability even when external inputs are reduced.
Mining and widespread use of phosphate fertilisers can add large amounts of phosphorus to soils and waters.
This can shift which nutrient is limiting and alter ecosystem productivity patterns.
Practice Questions
State one reason the phosphorus cycle lacks an atmospheric phase and one consequence of this for ecosystems. (2 marks)
1 mark: Explains lack of atmospheric phase (e.g., phosphorus is not volatile / exists mainly as phosphate in rocks and particles, not as gases).
1 mark: Consequence (e.g., slower cycling and redistribution / phosphorus becomes scarce and can limit productivity).
In an undisturbed ecosystem, explain why phosphorus is often the limiting nutrient. In your answer, refer to phosphorus storage forms and the processes that add and remove biologically available phosphate. (5 marks)
1 mark: Identifies main storage in rocks/sediments/soil particles (solid mineral forms).
1 mark: Explains no atmospheric reservoir so no rapid global replenishment/mixing.
1 mark: Adds phosphate mainly via slow weathering (input is slow).
1 mark: Removes available phosphate via runoff/erosion and/or burial in sediments (losses can be long-term).
1 mark: Links low availability to constrained growth/primary productivity (definition/application of limiting nutrient).

