AP Syllabus focus:
‘Phospholipids have hydrophilic heads facing aqueous environments and hydrophobic tails facing inward, forming the bilayer core of plasma membranes.’
Cell membranes are built around phospholipids whose chemistry makes them self-assemble in water. Understanding the bilayer’s structure explains why membranes are stable barriers yet flexible enough for cellular life.
Amphipathic phospholipids: the building blocks
Phospholipid architecture and polarity
A phospholipid consists of:
3D space-filling model of a saturated fatty acid, emphasizing the long nonpolar hydrocarbon chain that makes lipid tails hydrophobic. This visualization helps connect tail structure to tight packing and van der Waals interactions within the membrane’s interior. Source
A phosphate-containing head that is polar (often charged) and mixes well with water
Two fatty acid tails that are nonpolar and avoid water
This dual nature makes phospholipids amphipathic.
Amphipathic: Having both hydrophilic (water-attracting) and hydrophobic (water-repelling) regions in the same molecule.
Because the head interacts favorably with water and the tails do not, phospholipids orient in consistent ways at water–lipid interfaces.
Hydrophilic heads face water
In cells, water is present on both sides of the plasma membrane:
Extracellular fluid outside the cell
Cytosol inside the cell
Therefore, the hydrophilic heads face outward toward these aqueous environments. The heads can form electrostatic interactions and hydrogen bonds with water, stabilising this orientation.
Hydrophobic tails face inward
The hydrophobic tails cluster away from water, facing inward toward one another. This creates a hydrophobic interior that forms the membrane’s core, acting as a barrier to many water-soluble substances.
The phospholipid bilayer: how structure emerges in water
Self-assembly into a bilayer
When many phospholipids are placed in water, they spontaneously organise so that:
Heads contact water
Tails are sequestered from water
In a typical cell membrane, this produces a bilayer—two layers of phospholipids arranged tail-to-tail.

Cross-section of a phospholipid bilayer showing the aqueous environments on both sides (extracellular vs intracellular) and the consistent orientation of molecules. The diagram labels hydrophilic (polar) heads facing water and hydrophobic tails packed inward, highlighting how the nonpolar core forms a barrier. Source
Phospholipid bilayer: A double layer of phospholipids in which hydrophilic heads face aqueous environments and hydrophobic tails face inward, forming the core structure of cellular membranes.
This matches the syllabus statement directly: hydrophilic heads facing aqueous environments and hydrophobic tails facing inward creates the bilayer core of plasma membranes.
Why bilayers (not single layers) dominate
A single layer would expose hydrophobic tails to water on one side. A bilayer solves this by burying tails between two layers, minimising energetically unfavorable water–tail contact. This is a central reason membranes form closed, continuous boundaries around cells and compartments.
Membrane thickness and internal environment
The inward-facing tails create a thin nonpolar region. This interior is critical because it separates the cell’s aqueous cytosol from the aqueous exterior, establishing a structural basis for controlled exchange and internal conditions.
Orientation, interactions, and stability
Head group interactions
Hydrophilic heads contribute to membrane stability via:
Hydrogen bonding with surrounding water
Electrostatic interactions among water, ions, and charged head groups
These interactions help keep the bilayer positioned at the interface between two water-based solutions.
Tail interactions and hydrophobic effect
The tails stabilise the bilayer by:
Clustering together to reduce exposure to water
Engaging in van der Waals interactions with neighbouring tails
The tails’ tendency to avoid water is a major driver of bilayer formation.
Hydrophobic effect: The tendency of nonpolar substances to aggregate in aqueous solutions, reducing contact with water.
A membrane is stable not because tails “attract” water away, but because the system lowers free energy when nonpolar tails are shielded from water.
Structural variability within phospholipids
Saturated vs unsaturated tails
Phospholipids differ in their fatty acid tails:
Saturated tails have no double bonds and are relatively straight, allowing tighter packing
Unsaturated tails contain one or more double bonds that introduce kinks, reducing packing efficiency
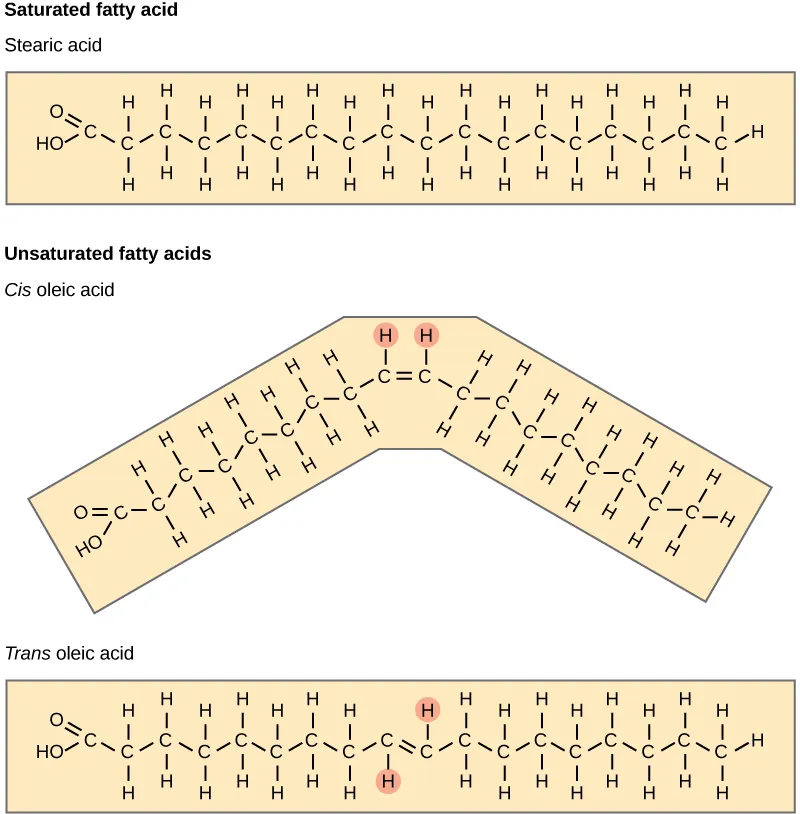
Comparison of saturated and unsaturated fatty acids showing how bond type changes tail geometry. The cis double bond creates a pronounced kink, while saturated chains remain straight, illustrating why unsaturated tails pack less tightly in membrane interiors. Source
These structural differences influence how tightly the bilayer’s interior can pack, affecting flexibility and the ease with which lipids move within the membrane plane.
Diversity of head groups
Different phospholipids can have different head groups, altering:
Net charge and polarity at the membrane surface
Interactions with water and dissolved ions
Even though AP Biology often focuses on the bilayer’s basic arrangement, recognising that head groups vary helps explain why different membranes can have distinct properties while maintaining the same core structure.
Spatial organisation: what “faces outward” means
Two aqueous faces, one hydrophobic core
A phospholipid bilayer has three key regions:
Outer surface: hydrophilic heads facing extracellular fluid
Inner surface: hydrophilic heads facing cytosol
Middle core: hydrophobic tails facing inward
This organisation creates a boundary that is chemically different at the surfaces versus the core, which is foundational for how membranes behave as selective barriers.
Continuous, self-sealing boundaries
If a small tear forms, phospholipids tend to rearrange so tails are not exposed to water, which promotes resealing. This property supports membrane integrity during normal cellular movements and shape changes.
FAQ
Bilayers are favoured when phospholipids have two fatty acid tails and a roughly cylindrical shape, which packs well into sheets.
Micelles are more common with single-tailed, cone-shaped lipids (e.g., some detergents), which curve strongly to form spheres.
Higher temperature increases lipid movement, reducing packing and making the bilayer more flexible.
Lower temperature decreases movement, increasing packing and making the bilayer more rigid; some membranes undergo a phase transition from more fluid to more gel-like states.
Lateral movement is diffusion within the same leaflet and happens readily.
Flip-flop (movement between leaflets) is rare without enzymes because a polar head would have to pass through the hydrophobic core; specialised enzymes (flippases/scramblases) can facilitate it.
Often they do not. Many membranes are asymmetric, with different head groups enriched on the cytosolic versus extracellular leaflet.
This asymmetry can influence surface charge, curvature tendencies, and how the membrane interacts with other molecules.
Curvature can result from differences in lipid shapes and sizes between leaflets.
Factors include:
Enrichment of cone-shaped lipids in one leaflet
Localised packing differences
Physical constraints from the surrounding cellular architecture
Practice Questions
Describe the arrangement of phospholipids in a plasma membrane bilayer. (2 marks)
Hydrophilic (polar) heads face the aqueous environments (cytosol and extracellular fluid). (1)
Hydrophobic (nonpolar) tails face inward towards each other, forming the bilayer core. (1)
Explain how the amphipathic nature of phospholipids leads to the formation and stability of a bilayer in water. (6 marks)
Phospholipids are amphipathic, with hydrophilic heads and hydrophobic tails. (1)
Hydrophilic heads form interactions with water (e.g., hydrogen bonding/electrostatic interactions). (1)
Hydrophobic tails avoid contact with water. (1)
Tails cluster together, reducing exposure of nonpolar regions to water (hydrophobic effect). (1)
This produces a tail-to-tail bilayer with heads facing the aqueous solutions on both sides. (1)
Bilayer is stable because it minimises energetically unfavourable water–tail contact and is self-sealing if tails become exposed. (1)

