AP Syllabus focus: ‘Use Lewis diagrams and VSEPR together to predict geometry, bond angles, relative bond energies (bond order), relative bond lengths (multiple bonds and atomic radius), and dipole moments.’
Molecular structure is a model that links electron arrangement to observable properties. By combining Lewis diagrams with VSEPR, you can predict 3-D geometry, approximate bond angles, and trends in bond length, bond energy, and polarity.
Connecting Lewis Structures to 3-D Shape
What each model contributes
Lewis diagram: shows connectivity (which atoms are bonded) plus lone pairs and multiple bonds on specific atoms.
VSEPR theory: uses electron-domain repulsions (bonding regions and lone pairs) to predict the 3-D arrangement around a central atom.
Stepwise workflow (AP-appropriate)
Draw the correct Lewis structure (including lone pairs and multiple bonds).
Identify the central atom’s electron domains:
Each single bond, double bond, or triple bond counts as one electron domain.
Each lone pair counts as one electron domain.
Use electron-domain count to determine the electron-domain geometry (the idealized arrangement).
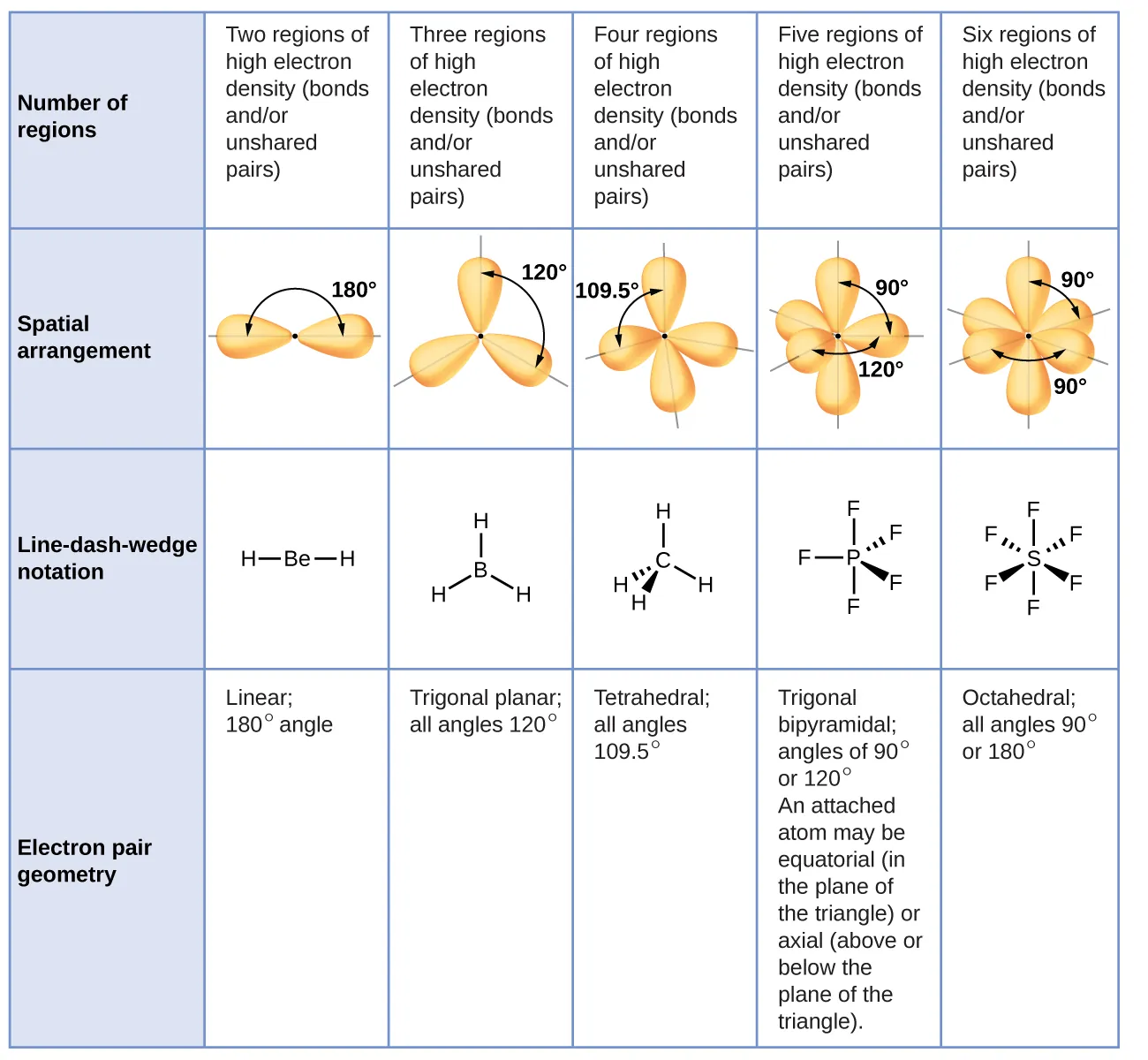
This table shows the ideal electron-domain geometries predicted by VSEPR for 2–6 regions of electron density and the corresponding ideal bond angles. It provides the baseline (linear, trigonal planar, tetrahedral, trigonal bipyramidal, octahedral) used before accounting for lone-pair compressions or multiple-bond repulsion effects. Source
Then determine the molecular geometry (positions of atoms only), accounting for lone pairs.
Use the geometry to infer bond angles and whether individual bond dipoles cancel or add to a net molecular dipole.
Predicting Bond Angles
Bond angles come from the best spatial separation of electron domains, but real molecules deviate from ideal angles when repulsions are unequal.
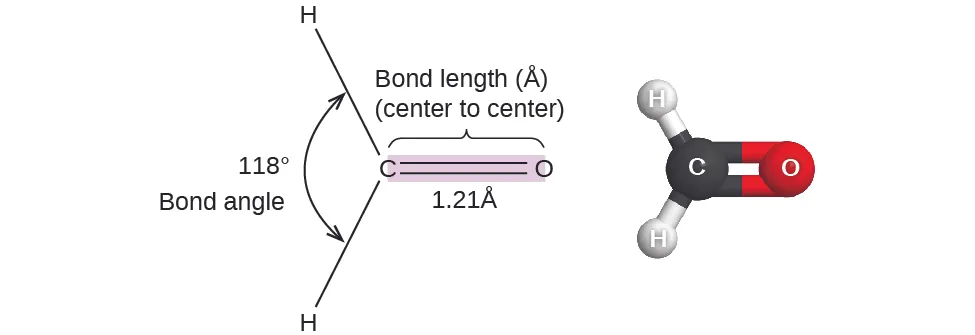
This figure labels both bond angles and a bond length for formaldehyde (HCO), pairing a structural drawing with a ball-and-stick model. It reinforces how geometry is quantified experimentally (angles and distances) and why VSEPR predictions are best treated as approximate when different electron domains (e.g., a double bond vs single bonds) distort ideal angles. Source
Ideal angle trends (qualitative use):
More domains generally means smaller ideal angles (e.g., 2 domains larger than 4 domains).
Lone pair effects:
Lone pairs repel more strongly than bonding pairs, so they compress adjacent bond angles.
More lone pairs on the central atom typically means smaller bond angles between atoms.
Multiple-bond effects (within VSEPR’s domain model):
A multiple bond is one domain, but it often exerts a stronger repulsive influence than a single bond, slightly adjusting nearby angles.
Use predicted angles as approximate values unless specific values are given; AP questions often ask for relative comparisons (larger/smaller) rather than exact numbers.
Predicting Relative Bond Lengths and Bond Energies
Lewis structures encode bond order, which strongly affects both bond length and bond energy.
Bond order: the number of shared electron pairs between two atoms (single = 1, double = 2, triple = 3).
Between two definition-level ideas, remember that geometry and bond properties are connected but not identical: VSEPR gives arrangement, while bond order and atom size set bond strength and distance.
Bond length trends:
Higher bond order → shorter bonds (triple < double < single).
Larger atomic radius (down a group) → longer bonds for the same bond order because bonding occurs between larger electron clouds.
Bond energy trends (relative, not numerical unless provided):
Higher bond order → greater bond energy (triple > double > single) because more electron density is shared between nuclei.
Shorter bonds are generally stronger because nuclei are held together more tightly by shared electrons (qualitative connection).
When comparing species, always compare the same pair of atoms if possible (e.g., C–C vs C–C); changing elements changes both size and electronegativity, complicating trends.
Predicting Molecular Polarity (Dipole Moments)
Bond polarity comes from electronegativity differences, but molecular polarity depends on the vector sum of all bond dipoles and lone-pair effects in 3-D.
Dipole moment: a measure of charge separation in a bond or molecule; direction points toward the more electronegative region and magnitude increases with greater separation of partial charges.
To decide if a molecule has a net dipole:
Determine which bonds are polar (qualitatively).
Use VSEPR-predicted shape to see whether bond dipoles cancel (symmetry) or reinforce.
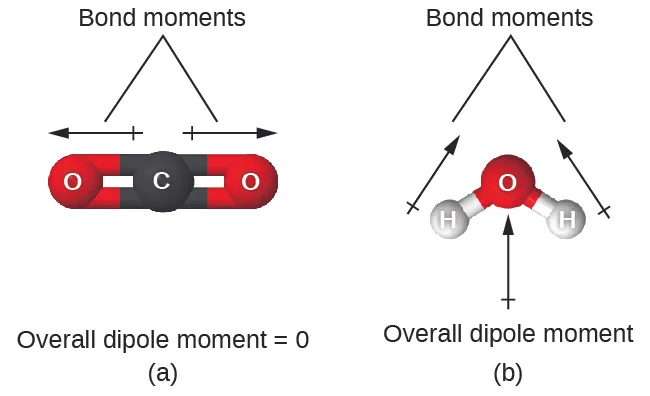
These diagrams compare a linear molecule (CO) where equal bond dipoles cancel to give zero net dipole with a bent molecule (HO) where bond dipoles add to produce a net molecular dipole. The visual emphasizes that polarity is a vector-sum property determined by geometry, not just by whether individual bonds are polar. Source
Key idea: even with polar bonds, a highly symmetrical geometry can yield no net dipole, while asymmetric arrangements (often due to lone pairs or different surrounding atoms) commonly produce a net dipole.
Be cautious to distinguish:
Polar bonds (bond-level property)
Polar molecules (shape-dependent, whole-molecule property)
FAQ
Look for symmetry in the VSEPR shape and identical surrounding atoms.
Useful cues:
Same outer atoms + symmetric geometry → often cancels
Lone pairs on the central atom → often breaks symmetry
Yes. The net dipole can be zero if the bond dipoles are equal in magnitude and arranged symmetrically so their vector sum is zero.
A lone pair is attracted to only one nucleus, so its electron density is more concentrated near the central atom, creating stronger repulsion that pushes bonding domains closer together.
Use this order of checks:
Compare bond order first (3 < 2 < 1 in length).
If bond order is the same, compare atomic radii (larger atoms form longer bonds).
No. VSEPR gives reliable qualitative structure and approximate angles; dipole moments are predicted as direction/magnitude trends from shape and bond polarity, not precise numerical values.
Practice Questions
(1–3 marks) A student draws a correct Lewis structure for showing one lone pair on S and two S–O bonding regions. Using VSEPR, state (i) the molecular geometry and (ii) whether has a net dipole moment.
(i) Bent (or V-shaped) (1)
(ii) Has a net dipole moment / is polar (1)
Reason linked to non-cancelling bond dipoles due to bent shape (1)
(4–6 marks) Using Lewis structures and VSEPR only, compare and in terms of (a) molecular geometry, (b) relative bond angles, (c) presence/absence of a net dipole moment, and (d) relative C–O vs S–O bond lengths assuming comparable bond order is shown in each Lewis structure.
(a) linear; bent (2)
(b) bond angle and larger than (1)
(c) non-polar (dipoles cancel); polar (dipoles do not cancel) (2)
(d) S–O longer than C–O due to larger atomic radius of S (1)

