AP Syllabus focus: ‘Use hybridization terms to describe electron arrangement around a central atom: sp gives 180°, sp² gives 120°, and sp³ gives 109.5° ideal bond angles.’
Hybridization is a naming tool that links a central atom’s electron-domain arrangement to a simple geometry and an ideal bond angle. On the AP Exam, you use sp, sp², and sp³ to communicate these idealised angles.
What hybridization means (for AP Chemistry)
Hybridization is used as a concise label for the electron-domain geometry around a central atom, based on how many regions of electron density surround it.
Hybridization: A model-based label describing the mixing of atomic orbitals on a central atom to produce a set of orbitals oriented for a particular electron-domain geometry (e.g., sp, sp², sp³).
In AP Chemistry, you typically infer the hybridization from the number of electron domains (not from drawing orbitals).
Electron domain: A region of electron density around a central atom; each single bond, multiple bond (counts as one domain), or lone pair counts as one domain.
Because this subtopic focuses on ideal bond angles, the key is matching the domain count to an ideal geometry and angle.
The required hybridization labels and ideal bond angles
sp: 2 electron domains → 180°
Hybridization: sp
Electron-domain geometry: linear
Ideal bond angle: 180°
Interpreting the label: “sp” corresponds to two equivalent directions of electron density, pointing opposite each other.
sp²: 3 electron domains → 120°
Hybridization: sp²
Electron-domain geometry: trigonal planar
Ideal bond angle: 120°
Interpreting the label: “sp²” corresponds to three equivalent directions in one plane, spaced evenly.
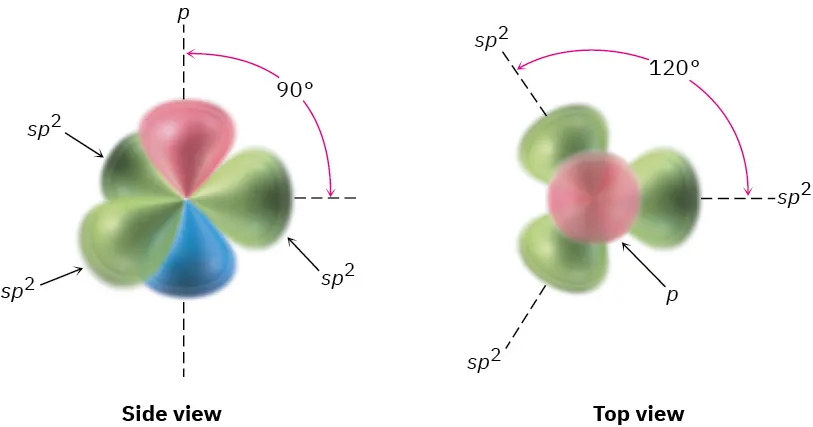
The figure on this page depicts three coplanar hybrid orbitals separated by ideal 120° angles, with an unhybridized orbital perpendicular to the plane. This provides a concrete visual link between “3 electron domains → trigonal planar” and the 120° ideal bond angle. Source
sp³: 4 electron domains → 109.5°
Hybridization: sp³
Electron-domain geometry: tetrahedral
Ideal bond angle: 109.5°

This diagram shows the tetrahedral spatial orientation of four equivalent hybrid orbitals. It emphasizes that the electron-domain geometry for four electron domains is tetrahedral, with an ideal angle of about 109.5° between directions of electron density. Source
Interpreting the label: “sp³” corresponds to four equivalent directions pointing toward the corners of a tetrahedron.
How to assign sp, sp², or sp³ quickly
Step-by-step rules (domain counting)
Identify the central atom (usually the least electronegative, never hydrogen).
Count electron domains around that atom:
Each single bond = 1 domain
Each double bond = 1 domain (still one region of electron density)
Each triple bond = 1 domain
Each lone pair = 1 domain
Assign hybridization from the total:
2 domains → sp → 180°
3 domains → sp² → 120°
4 domains → sp³ → 109.5°
“Ideal” vs real bond angles (what to report)
The angles above are ideal bond angles for the electron-domain geometry. Real molecules can deviate, but for this syllabus point you should know and state:
sp = 180°
sp² = 120°
sp³ = 109.5°
Common pitfalls to avoid
Don’t let bond order change the domain count: a multiple bond still counts as one electron domain.
Don’t confuse molecular shape with the electron-domain geometry label; hybridization is tied to the domain geometry and its ideal angle.
Don’t assign hybridization to terminal atoms (like H) in typical AP tasks; the focus is the central atom’s arrangement.
FAQ
Hybridisation uses regions of electron density. A double bond has higher density than a single bond, but it is still one directionally localised region around the central atom.
Use “$sp$” when asked for hybridisation, and “linear” when asked for electron-domain geometry. If a prompt is vague, include both alongside the ideal angle.
Yes. Lone pairs are electron domains. Count them exactly like bonds when determining the domain total for hybridisation naming.
Bond angles near $109.5^\circ$ (from diffraction/spectroscopy) support a tetrahedral electron-domain arrangement, which corresponds to an $sp^3$ label in the model.
In the ideal model, $sp^2$ implies trigonal planar electron-domain geometry (planar). Apparent non-planarity can occur due to larger structural constraints, but the hybridisation label still reflects the local domain arrangement.
Practice Questions
(2 marks) Determine the hybridisation of the central atom and state the ideal bond angle for a species with 3 electron domains around the central atom.
States (1)
States ideal bond angle (1)
(5 marks) For the central atom in each case below, (i) state the number of electron domains, (ii) give the hybridisation, and (iii) state the ideal bond angle:
A: 2 single bonds and 1 lone pair.
B: 1 double bond, 1 single bond, and 1 lone pair.
A: identifies 3 electron domains (1); (1); (1)
B: identifies 3 electron domains (double bond counts as one domain) (1); and (1)

