AP Syllabus focus: ‘A pi bond restricts rotation about a multiple bond, which can lead to geometric (cis/trans) isomers; connect bonding to observable differences between isomers.’
Geometric isomerism is one of the clearest ways bonding affects molecular behavior. Understanding why double bonds do not freely rotate helps explain when distinct, isolable isomers can exist and differ in properties.
Why bonds restrict rotation
A carbon–carbon double bond (and many other multiple bonds) consists of one sigma () bond and one pi () bond. The key idea is that forming and maintaining a bond requires a specific orbital alignment.
Pi () bond: a covalent bond formed by side-by-side overlap of parallel orbitals above and below the internuclear axis, adding electron density outside the line connecting the nuclei.
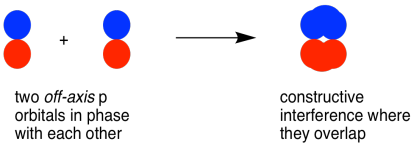
Side-by-side overlap of parallel p orbitals produces a bonding interaction, placing electron density above and below the internuclear axis. This visualization reinforces why maintaining a bond requires specific orbital alignment rather than arbitrary rotation. Source
Because overlap depends on parallel orbitals, rotating one atom relative to the other would misalign the orbitals and destroy the overlap.
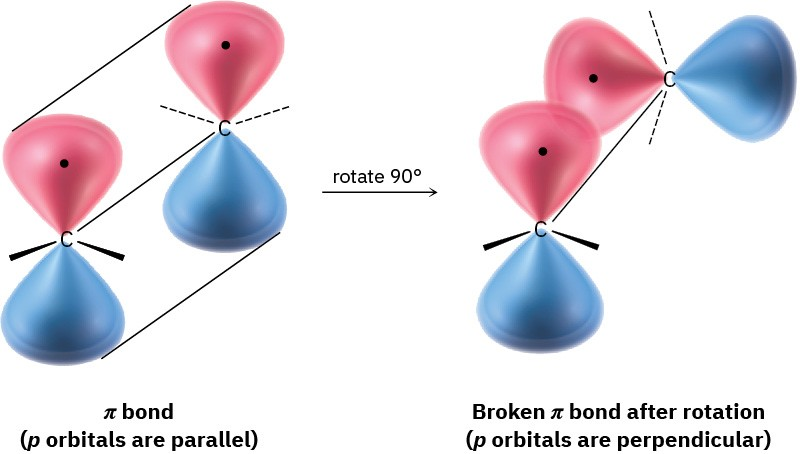
This diagram illustrates that rotation about a double bond is not a simple twist: the bond must be broken and then re-formed. That mechanistic picture explains why double-bond rotation has a large energy barrier compared with rotation about a single bond. Source
As a result:
Rotation about a single bond is usually relatively free (bond can twist without breaking).
Rotation about a double bond is restricted because rotation would require breaking the bond (a high-energy process).
This restricted rotation means different spatial arrangements around the double bond can be “locked in,” creating distinct compounds rather than rapidly interconverting conformations.
What geometric (cis/trans) isomerism means
Restricted rotation only matters for isomerism when there are different possible arrangements that cannot interconvert without breaking the bond.
Geometric (cis/trans) isomers: stereoisomers that differ in the spatial arrangement of substituents around a rigid unit (typically a double bond), producing distinct “same side” (cis) and “opposite side” (trans) forms.
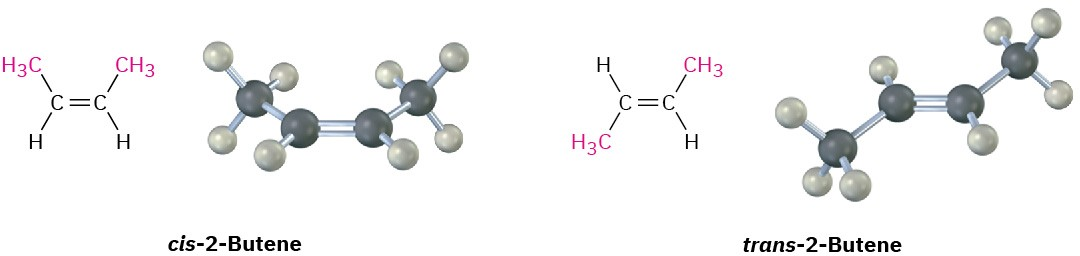
Cis- and trans-2-butene are shown as distinct, isolable stereoisomers with substituents on the same side versus opposite sides of the bond. The diagram connects “locked” substituent positions to the idea that interconversion would require disrupting the bond. Source
Geometric isomerism for alkenes occurs when each carbon of the has two different substituents. If one carbon has two identical groups, swapping “sides” produces the same structure, so no cis/trans pair exists.
Criteria for cis/trans isomerism in alkenes
Use these checks:
Confirm a multiple bond (commonly ) is present, creating restricted rotation.
On the left alkene carbon, verify the two attached groups are not identical.
On the right alkene carbon, verify the two attached groups are not identical.
If both carbons meet this requirement, two geometric isomers are possible.
Assigning cis vs trans (AP-level convention)
For many AP Chemistry contexts, cis/trans is described using matching groups:
cis: the identical or most comparable substituents are on the same side of the double bond.
trans: those substituents are on opposite sides.
When there is no obvious pair of matching substituents, chemists often use E/Z notation; however, the essential AP idea remains the same: restricted rotation around the bond can create two distinct arrangements.
Connecting bonding to observable differences between isomers
Because geometric isomers have different 3-D arrangements, they can differ in measurable properties even though they have the same molecular formula and the same connectivity.
Key links from structure to properties include:
Polarity and dipole moment
In some cis isomers, bond dipoles can add to create a net molecular dipole.
In some trans isomers, dipoles can partially cancel, lowering the net dipole.
Intermolecular forces
More polar isomers often exhibit stronger dipole–dipole attractions, influencing boiling points and solubility trends.
Packing and physical properties
A more symmetrical isomer may pack more efficiently in a solid, sometimes increasing melting point relative to a less symmetrical isomer.
These differences arise specifically because the bond locks substituents into fixed relative positions, making each geometric isomer a distinct substance with its own set of characteristic properties.
FAQ
It is typically far larger than $RT$ at room temperature, so spontaneous rotation is negligible.
That is why cis/trans forms are usually isolable rather than rapidly interconverting.
Yes. Rings restrict rotation because the ring framework is rigid.
Substituents can be locked on the same face (cis) or opposite faces (trans), creating distinct stereoisomers.
cis/trans becomes ambiguous when there is no clear pair of identical substituents to compare.
E/Z uses priority rules to assign a unique description for each arrangement.
They can have different symmetry and dipole changes during vibrations, affecting IR peak intensities.
They can also show different NMR chemical shifts and coupling patterns because neighbouring environments differ.
Yes. UV light can promote an electron to an excited state, weakening the $\pi$ bond and allowing rotation.
Certain catalysts can enable isomerisation pathways that effectively interchange cis and trans without simple thermal rotation.
Practice Questions
(2 marks) Explain why rotation about a double bond is restricted compared with rotation about a single bond.
Mentions that a double bond contains a bond formed by side-on overlap (1)
States rotation would break/misalign the overlap (requires breaking the bond, high energy), so rotation is restricted (1)
(5 marks) A student claims that any molecule containing a bond will have cis/trans isomers. Evaluate this claim. In your answer, state the condition required for geometric isomerism about a bond and describe one observable property difference that can result between the isomers.
Claim is not always true (1)
States requirement: restricted rotation about due to bond (1)
States correct structural condition: each alkene carbon must have two different substituents (2)
Gives one valid observable difference linked to structure (e.g., different polarity/dipole moment leading to different boiling point/solubility; or different packing leading to different melting point) (1)

