AP Syllabus focus: ‘Bond formation involves orbital overlap; multiple bonds contain one sigma bond and one or more pi bonds. Sigma overlap is stronger, giving sigma bonds greater bond energy than pi bonds.’
Bonding models become more predictive when you connect Lewis structures to how orbitals physically overlap in space. This page focuses on sigma (σ) and pi (π) bonds, their formation, and why they differ in strength.
Orbital overlap as the basis of covalent bonding
Covalent bonds form when electron density is concentrated between two nuclei due to the overlap of atomic (or hybrid) orbitals. Greater overlap generally means more electron density between nuclei, stronger attraction, and a more stable bond.
Orbital overlap: the spatial intersection of two orbitals that allows electron density to be shared between two atoms, stabilising the atoms as a covalent bond forms.
Overlap depends on orbital orientation and shape. Orbitals that point directly at each other can overlap more effectively than orbitals that meet side-by-side.
Sigma (σ) bonds: head-on overlap
A sigma bond forms from end-to-end (head-on) overlap along the internuclear axis (the line connecting the two nuclei). Because the overlap is concentrated directly between the nuclei, sigma bonds typically maximise electron density in the bonding region.
Sigma (σ) bond: a covalent bond formed by head-on orbital overlap with electron density concentrated along the internuclear axis.
Key features of σ bonds:
Present in every single bond (a single bond is one σ bond).
Formed by overlap such as:
–, –, – (when aligned head-on)
hybrid–hybrid overlaps (common in molecular structures)
Electron density has cylindrical symmetry around the bond axis (rotation about the axis does not change the σ bonding pattern).
Pi (π) bonds: side-by-side overlap
A pi bond forms from side-by-side overlap of two parallel unhybridized orbitals (in the AP model).
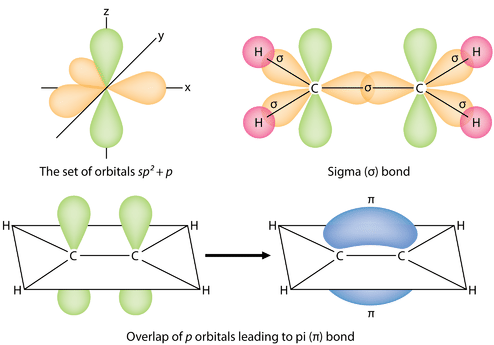
Diagram showing how a \sigma bond forms by head-on overlap along the internuclear axis, while a \pi bond forms by side-by-side overlap of parallel orbitals. The visual emphasizes that \pi electron density lies above and below the bond axis rather than directly between the nuclei, helping explain why \sigma overlap is typically stronger. Source
The overlap occurs above and below (or in front of and behind) the internuclear axis rather than directly on it.
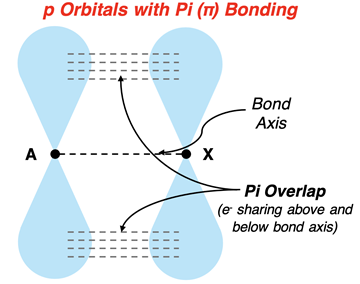
Visual model of a \pi bond formed by lateral overlap of unhybridized orbitals, showing electron density concentrated in regions above and below the internuclear axis. This layout reinforces why rotation can disrupt \pi overlap and why \pi bonding is usually weaker than head-on \sigma overlap. Source
Pi (π) bond: a covalent bond formed by side-by-side overlap of parallel orbitals, with electron density concentrated in two lobes on either side of the internuclear axis.
Key features of π bonds:
Occur only in multiple bonds.
Require proper alignment of the two parallel orbitals; misalignment reduces overlap and weakens the π interaction.
Electron density is not centered directly between nuclei, so the stabilising overlap is typically less effective than in σ bonding.
Multiple bonds: one σ plus one or more π
A multiple bond contains:
Double bond: 1 σ + 1 π
Triple bond: 1 σ + 2 π
This structure arises because only one head-on overlap can occur along a given internuclear axis between two atoms (making one σ bond). Any additional bonding must come from side-by-side overlap, producing π bonds.
When interpreting a Lewis structure with a double or triple bond, connect it to this overlap picture:
The first bond drawn between the atoms corresponds to the σ bond framework.

Ethene is modeled with one \sigma bond along the internuclear axis and a \pi bond formed by overlap above and below the plane of the molecule. The diagram makes the “one \sigma plus one \pi” structure of a double bond visually explicit, bridging Lewis structures and orbital-overlap reasoning. Source
The extra bond(s) correspond to π bond(s) that supplement the σ bond.
Why σ bonds have greater bond energy than π bonds
The syllabus emphasises that sigma overlap is stronger, giving σ bonds greater bond energy than π bonds. The core reason is overlap effectiveness:
σ bonds: head-on overlap places electron density directly between nuclei, maximising electrostatic attraction between nuclei and shared electrons.
π bonds: side-by-side overlap is less direct; electron density sits off the axis, so the overlap is usually smaller and the bond contribution is weaker.
Practical implications for AP-level reasoning:
Breaking a π bond generally requires less energy than breaking a σ bond (all else equal).
In a multiple bond, the σ bond is often treated as the “base” bond, with π bond(s) adding bonding but contributing less per bond than the σ.
Using σ/π bonding to describe structure
When asked to “count” σ and π bonds, use these rules:
Every single bond = 1 σ
Every double bond = 1 σ + 1 π
Every triple bond = 1 σ + 2 π
This σ/π language is also how you translate between structural drawings and an orbital-overlap explanation of bond strength and bonding capacity.
FAQ
π bonding depends strongly on parallel alignment; twisting reduces side-on overlap rapidly.
σ overlap is less sensitive because head-on overlap can remain substantial.
A π bond has a nodal plane containing the internuclear axis where electron density is zero.
This reflects the side-by-side phase relationship of the overlapping orbitals.
Yes; any two orbitals with appropriate symmetry can overlap side-by-side to form π bonding.
At AP level, π bonds are typically modelled as p–p overlap.
Only one head-on overlap can occupy the internuclear axis between the same two atoms.
Additional bonding must use different spatial regions, producing π bonds.
σ: electron density concentrated along the bond axis.
π: electron density concentrated in two regions on opposite sides of the axis.
Practice Questions
Ethene, C2H4, contains a carbon–carbon double bond. State how many σ bonds and how many π bonds are present in the molecule.
Correct total σ bonds (5) (1)
Correct total π bonds (1) (1)
Explain, in terms of orbital overlap, (i) how a double bond is formed between two atoms and (ii) why a σ bond has greater bond energy than a π bond.
Double bond consists of one σ bond and one π bond (1)
σ bond formed by head-on overlap along the internuclear axis (1)
π bond formed by side-by-side overlap of parallel orbitals above/below the axis (1)
σ overlap is greater/more effective, concentrating electron density directly between nuclei (1)
Therefore σ bonding interaction is stronger and has higher bond energy than π (1)

