AP Syllabus focus: ‘You are not assessed on deriving or drawing hybrid orbitals or d‑orbital hybridization; for expanded electron domains, focus on predicting molecular shape, plus sigma–pi bonding and sp/sp²/sp³ nomenclature.’
This page clarifies what AP Chemistry expects you to know about hybridization and bonding models, and what is explicitly excluded, so you can focus studying on exam-relevant predictions and vocabulary.
What the AP Exam does and does not assess
Core idea: models are used for predictions
On the AP Exam, hybridization language is a naming and prediction tool, not an orbital-drawing exercise. You should treat it as a compact way to communicate electron arrangement and likely geometry, consistent with a Lewis structure and VSEPR-based reasoning.
Explicit exclusions (not tested)
You are not assessed on:
Deriving hybrid orbitals from atomic orbitals (e.g., mathematically or conceptually “mixing” and orbitals to generate hybrids)
Drawing hybrid orbitals (shapes, lobes, orbital diagrams for hybrids, or detailed overlap sketches)
-orbital hybridization schemes (e.g., , labels, “ participation” explanations)
If a question tempts you to justify a geometry by invoking orbitals, that line of reasoning is outside the assessed scope. Instead, lean on electron domains and molecular shape prediction.
What you are responsible for knowing
Hybridization nomenclature that is fair game
You should be able to use and interpret:
(idealized 180° arrangement)
(idealized 120° arrangement)
(idealized 109.5° arrangement)
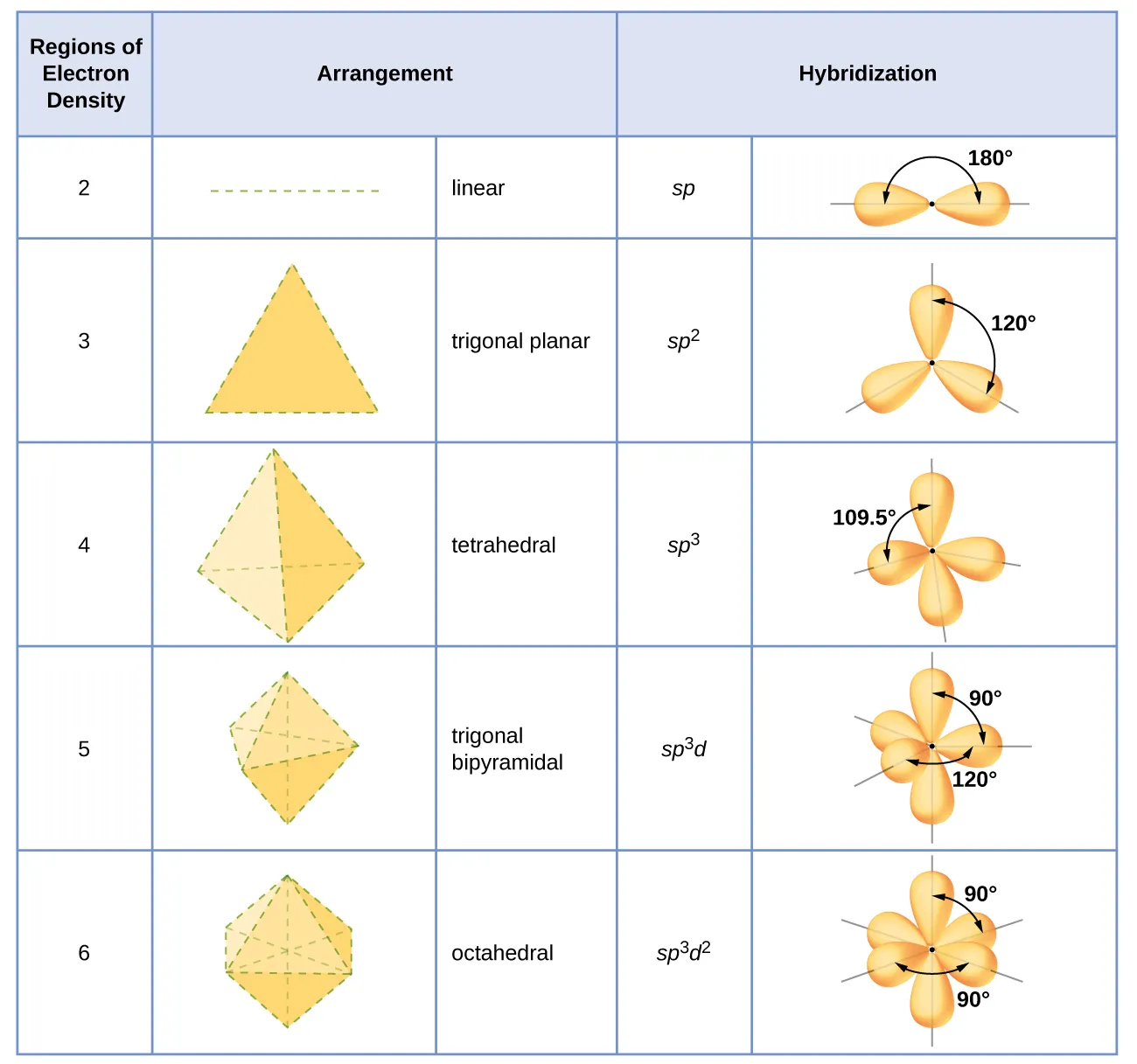
Table/diagram linking the number of electron-density regions to idealized geometries and the corresponding hybridization labels (, , , etc.), with the standard bond angles (180°, 120°, 109.5°). Use it as a quick “domains → geometry → vocabulary” reference when the prompt asks for hybridization nomenclature or shape prediction. (For AP Chem, focus on the , , and rows unless the question explicitly shifts to expanded-domain VSEPR shapes.) Source
These labels are used to describe the electron arrangement around a central atom in common cases. On the exam, this typically means connecting a correct Lewis structure to the number of electron domains and then selecting the appropriate nomenclature when applicable.
Expanded electron domains: what to do instead of -hybridization
For species with expanded electron domains (central atoms with more than four electron domains), your task is to:
Predict molecular shape using electron-domain reasoning (VSEPR-style logic)
Describe bonding in terms of sigma–pi bonding where multiple bonds are present
Use nomenclature only where it clearly applies; do not invent or rely on -hybridization labels to justify shapes
In other words, expanded-domain questions are assessed through shape prediction and bonding descriptions, not through hybrid-orbital derivations.
Sigma and pi bonding: what you must be able to state
When multiple bonds appear in a valid structure, you are expected to connect them to sigma and pi bonding in a qualitative, model-based way:
A single bond is a sigma bond
A double bond contains one sigma and one pi
A triple bond contains one sigma and two pi
You do not need to draw orbital overlap diagrams; you do need to correctly count and classify bonds as sigma vs. pi when asked.
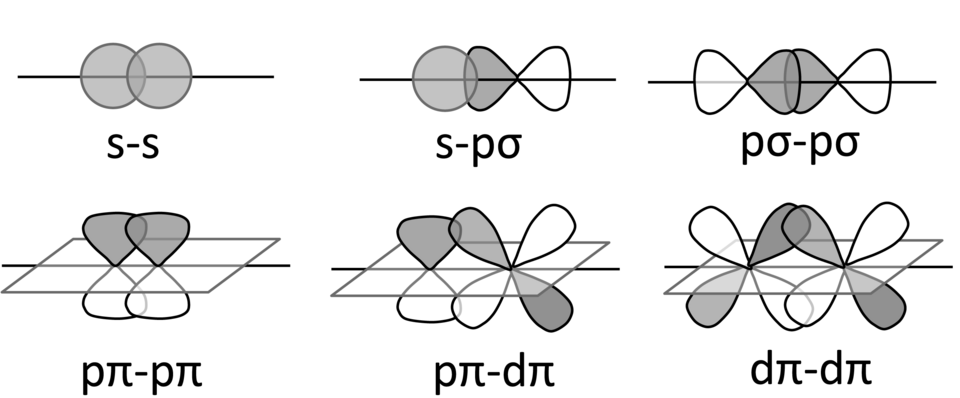
High-resolution schematic summarizing common (end-on) versus (side-on) overlap patterns for atomic orbitals, providing a conceptual picture of why bonding appears “above/below” the internuclear axis. This supports qualitative reasoning about why a double bond has one framework plus an added interaction, without requiring you to reproduce orbital drawings on an AP response. Use it as a conceptual anchor, then default back to Lewis-structure bond counting for exam questions. Source
How exam wording signals the expected approach
“Determine the molecular geometry” or “predict the shape”
Use electron domains and lone-pair considerations to select the molecular shape. Avoid any explanation based on orbital hybridization.
“Identify the hybridization of the central atom”
Limit responses to , , or in contexts where that nomenclature matches the electron-domain arrangement being tested. If the structure has expanded domains, focus on shape (and bond types) rather than -based labels.
“How many sigma and pi bonds are present?”
Answer with correct sigma–pi counting from the bond types shown (single/double/triple), without orbital drawings.
High-utility pitfalls to avoid
Don’t lose points by adding unasked, off-scope claims like “ explains trigonal bipyramidal.” The exam prefers shape reasoning over -hybridization assertions.
Don’t conflate electron-domain arrangement with molecular shape; the exam may focus on shape even when electron domains differ due to lone pairs.
Don’t treat hybridization as something you must “prove” with orbital sketches; the assessed skill is consistent classification and prediction.
FAQ
Use molecular shape names and electron-domain reasoning (e.g., “five electron domains” plus the predicted shape). Avoid $d$-hybridisation labels in explanations.
Usually you won’t gain credit for it. If it replaces correct shape reasoning or introduces incorrect claims, it can cost clarity marks on explanations.
Answer with $sp$, $sp^2$, or $sp^3$ only when the electron-domain arrangement clearly matches. Otherwise, prioritise the geometry and bond descriptions the question asks for.
Describe the count of electron domains around the central atom and state that domains arrange to minimise repulsion, leading to the named molecular shape.
Yes. Many questions only require counting single/double/triple bonds to get sigma and pi totals; no hybridisation statement is needed unless explicitly requested.
Practice Questions
(2 marks) State two topics related to hybridisation that are explicitly not assessed on the AP Chemistry exam.
1 mark: States not assessed on deriving or drawing hybrid orbitals (either “deriving” or “drawing” accepted).
1 mark: States not assessed on -orbital hybridisation / or type schemes.
(5 marks) A student explains the shape of a molecule with five electron domains by claiming the central atom forms hybrid orbitals. (a) (2 marks) Explain why this justification is not required for AP Chemistry assessment. (b) (3 marks) State three exam-relevant things the student should focus on instead, for such a molecule.
1 mark: Identifies -orbital hybridisation is excluded from assessment.
1 mark: Identifies deriving/drawing hybrid orbitals is not assessed; emphasis is on prediction rather than orbital construction. (b) Any three, 1 mark each:
Predict molecular shape from electron domains (VSEPR-style).
Describe sigma vs. pi bonding where multiple bonds occur.
Use nomenclature only where applicable (not -based labels).

