AP Syllabus focus: ‘Trends across periods and down groups can be understood using the shell model and the ideas of shielding and effective nuclear charge.’
Shielding and effective nuclear charge explain why outer electrons are held more or less tightly in different atoms. Using a shell model and electrostatic ideas, you can justify major periodic patterns without memorising data.
The shell model and electron–nucleus attraction
In the shell model, electrons occupy principal energy levels ().
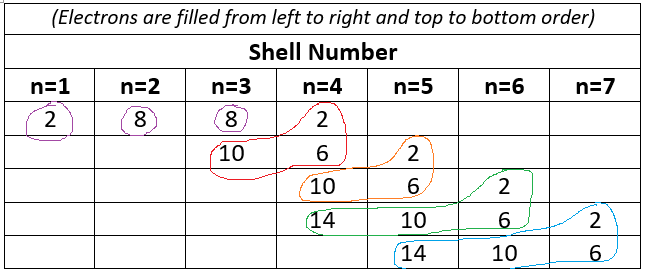
A compact chart showing how electrons fill principal shells ( through ) across the periodic table structure. This reinforces the shell-model idea that moving down a group introduces a new principal energy level (greater distance and more inner electrons). It provides structural context for why shielding changes dramatically when new shells are added. Source
Valence electrons (outer-shell electrons) experience two competing effects:
Attraction to the positively charged nucleus (more protons means stronger pull)
Repulsion from other electrons, especially core electrons, which reduces the pull felt by valence electrons
These ideas allow qualitative comparisons of how strongly an atom’s valence electrons are attracted to its nucleus.
Shielding
Core electrons reduce the nucleus–valence attraction by repelling valence electrons.
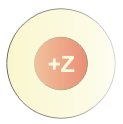
A schematic showing how core electrons surround the nucleus and block (screen) some of the nucleus’s positive charge from reaching valence electrons. The reduced visible “+” region illustrates why valence electrons experience a weaker net attraction than the full nuclear charge would suggest. This is a visual model of shielding rather than a literal picture of electron positions. Source
Shielding: The reduction in electrostatic attraction between the nucleus and a valence electron due to repulsion by other electrons (especially inner/core electrons).
Shielding is mainly determined by how many inner shells lie between the nucleus and the valence shell:
More occupied inner shells more shielding
Electrons in the same shell shield one another only partially, so shielding does not “cancel” nuclear attraction within a shell
Why shielding is a “shell” effect
Because core electrons are closer to the nucleus, they spend much of their time between the nucleus and valence electrons, strongly reducing the net attraction felt by valence electrons. This is why moving down a group (adding shells) changes electron–nucleus attraction so noticeably.
Effective nuclear charge ()
To describe the net pull felt by a valence electron, chemists use effective nuclear charge, which folds together nuclear charge and shielding.
Effective nuclear charge (): The net positive charge “felt” by an electron in an atom after accounting for shielding by other electrons.
A useful qualitative model treats as the nuclear charge minus a shielding amount.
= effective nuclear charge (unitless)
= atomic number (number of protons; unitless)
= shielding constant (how much nuclear charge is screened; unitless)
This equation is mainly conceptual in AP Chemistry: you typically compare qualitatively rather than computing precisely.
Trend across a period: increasing
Across a period (left to right):
increases by 1 each step (more protons)
Added electrons go into the same principal shell (same ), so the number of core shells stays the same
Shielding increases only slightly, because same-shell electrons do not shield very effectively
Therefore, increases across a period.
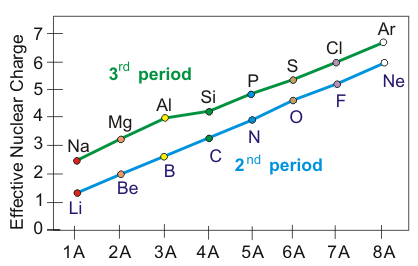
A plot of effective nuclear charge versus group position for the 2nd and 3rd periods, showing a clear rise in from left to right. Because the added electrons go into the same principal shell, shielding does not increase enough to offset the higher nuclear charge. The result is a stronger net pull on valence electrons across the period. Source
Interpreted with the shell model: valence electrons in the same shell are pulled more strongly toward a nucleus with more protons, because shielding does not rise enough to offset the increased nuclear charge.
Trend down a group: shielding increases, for valence electrons stays similar
Down a group:
Valence electrons occupy a higher (farther from the nucleus in the shell model)
Each step adds a new inner shell, greatly increasing shielding
increases, but much of that increased nuclear charge is offset by added core electrons
So, the experienced by valence electrons is often roughly similar within a group, while shielding and distance both increase. In qualitative reasoning, this is why atoms lower in a group do not hold their outer electrons as tightly as you might expect from alone.
How to justify comparisons (AP-style reasoning)
When asked to compare attractions or trends, explicitly reference:
Number of protons (): higher strengthens attraction
Shielding by core electrons: more inner shells increases shielding substantially
Same-shell shielding: increases modestly across a period
Shell model distance (higher ): valence electrons are farther and less strongly attracted, even if is larger
FAQ
No. Core electrons experience a much larger net attraction.
Valence electrons experience more shielding, so their $Z_\text{eff}$ is smaller.
Same-shell electrons have similar average distances from the nucleus, so they are less consistently “between” the nucleus and each other.
Shielding is strongest when electrons are closer to the nucleus than the electron of interest.
No. Slater’s rules are beyond typical AP expectations.
You should use $Z_\text{eff}=Z-S$ as a qualitative model to justify trends.
Their electron density is not as concentrated close to the nucleus as s electrons.
As a result, they are less effective at screening nuclear charge from outer electrons.
Not directly. It is inferred from experimental patterns (for example, energies needed to remove electrons).
Different models can estimate $Z_\text{eff}$, so it is best viewed as a useful conceptual quantity rather than a directly observed value.
Practice Questions
(2 marks) Define shielding and state how shielding changes from Li to Ne.
Correct definition of shielding as reduced nucleus–valence attraction due to electron–electron repulsion/core electrons (1).
Shielding changes only slightly across Li to Ne because electrons are added to the same shell; core shielding is essentially constant (1).
(5 marks) Using the shell model, compare the effective nuclear charge felt by a valence electron in Na and Mg, and then compare Na and K. Explain your reasoning.
Na vs Mg: states Mg has greater (more protons) (1).
Na vs Mg: states shielding by core electrons is similar (same core configuration) so is higher in Mg (1).
Na vs K: states K has an additional occupied shell (valence electron in higher ) (1).
Na vs K: states K has greater shielding due to extra core shell (1).
Concludes K valence electron feels similar or lower than Na (and is held less strongly) due to increased shielding/distance offsetting higher (1).

