AP Syllabus focus: ‘Predict and explain ionization energy trends using Coulomb’s law, electron shielding, and effective nuclear charge based on periodic-table position.’
Ionization energy trends are best understood as electrostatic attractions between the nucleus and valence electrons. Periodic patterns emerge because nuclear charge, distance from the nucleus, and shielding change predictably across periods and down groups.
Core idea: electrostatic attraction controls ionization energy
First ionization energy: The energy required to remove the outermost electron from a neutral atom in the gas phase, forming a ion.
A higher ionization energy means an electron is held more strongly. The strength of attraction depends on (1) how positive the nucleus effectively “feels” to that electron and (2) how far away the electron is.
= Electrostatic force (relative strength)
= Charges on interacting particles (relative magnitude)
= Distance between nucleus and electron (relative distance)
This Coulomb’s law relationship is used qualitatively: increasing nuclear charge tends to increase attraction, while increasing electron–nucleus distance decreases attraction strongly (inverse-square dependence).
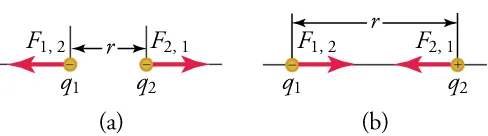
This diagram illustrates Coulomb’s law for two point charges separated by a distance , with force vectors shown on each charge. It emphasizes that the electrostatic force acts along the line joining the charges and that the direction depends on whether the charges are like (repulsive) or unlike (attractive). Source
Shielding and effective nuclear charge ()
Electrons do not experience the full nuclear charge because inner (core) electrons repel outer electrons, reducing nuclear attraction.
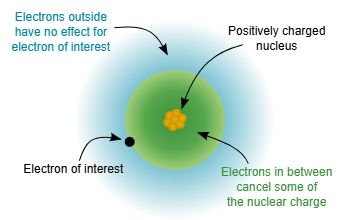
This schematic shows how core electrons between the nucleus and a valence electron reduce the net attractive pull experienced by the valence electron (electron shielding). The picture supports the idea that the valence electron responds to an effective nuclear charge, , rather than the full nuclear charge . Source
Shielding: Reduction in nuclear attraction for a valence electron due to repulsion by core electrons located between the nucleus and that valence electron.
Shielding is mainly determined by how many inner shells are present; electrons in the same shell shield relatively poorly compared with electrons in inner shells.

This figure highlights which electrons are classified as core versus valence in a core-abbreviated electron configuration. It helps connect shielding to electron configuration by showing that inner (core) electrons are treated separately from the outer (valence) electrons most directly involved in ionization. It also supports the idea that adding inner shells changes shielding substantially down a group. Source
Effective nuclear charge (): The net positive charge felt by an electron after accounting for shielding by other electrons.
As a rule, higher and smaller distance () both increase ionization energy because the electron is more strongly attracted to the nucleus.
Trend across a period (left to right): generally increases
Across a period, ionization energy generally increases because:
Nuclear charge increases (more protons), which increases attraction to electrons.
Added electrons go into the same principal energy level, so distance from the nucleus does not increase much.
Shielding changes little because the number of core electron shells stays the same.
Therefore increases, tightening the hold on valence electrons.
Common “dips” within a period (qualitative explanations)
Small decreases can occur even as rises, due to electron arrangement in subshells:
Subshell change (s to p): When the first electron enters a higher-energy p subshell, it is easier to remove than an s electron in the same shell (p is higher in energy and often slightly less penetrating).
Electron pairing in a p orbital: When electrons begin to pair in the same orbital, electron–electron repulsion increases, making removal slightly easier than from a half-filled set of orbitals.
These deviations are still explained using the same AP tools: Coulombic attraction (energy/penetration), shielding (similar), and effective nuclear charge (increasing but not the only factor).
Trend down a group (top to bottom): generally decreases
Down a group, ionization energy generally decreases because:
The valence electron is in a higher principal energy level (larger average distance ).
There are more inner shells, so shielding increases substantially.
Although nuclear charge increases, the combined effect of greater distance and greater shielding reduces the nucleus–electron attraction experienced by the valence electron (lower effective pull).
In Coulomb’s law terms, the increase in (and the shielding-driven decrease in effective attraction) outweighs the increase in nuclear charge, so less energy is needed to remove the valence electron.
How to justify comparisons using periodic-table position
When comparing two first ionization energies, structure your reasoning around:
Relative (often higher to the right, similar within a group trend context)
Relative distance () (larger down a group; similar across a period)
Relative shielding (much larger down a group; nearly constant across a period)
Any subshell or pairing difference that changes electron energy/repulsion
Use periodic-table position to identify which factor dominates, then state the direction of the effect on attraction and ionization energy.
FAQ
Half-filled orbitals minimise pairing, reducing electron–electron repulsion within the subshell.
With less repulsion, an electron is held more strongly, so more energy is needed to remove it.
A large jump between successive ionisation energies indicates removal has moved from valence to core electrons.
Core electrons are closer (smaller $r$) and experience much higher $Z_\text{eff}$, so they require far more energy.
Same-shell electrons have similar average distances and spend time at comparable radii, so they do not consistently sit “between” each other and the nucleus.
Inner-shell electrons provide the strongest shielding.
Tabulated ionisation energies refer to isolated gaseous atoms.
In compounds, interactions can change electron removal energetics, but that is not the definition of ionisation energy used for periodic trends.
Atoms in the gas phase are subjected to increasing energy (often via spectroscopy methods) to produce $1+$ ions.
The threshold energy required corresponds to the first ionisation energy under defined conditions.
Practice Questions
(2 marks) Using Coulomb’s law ideas, explain why the first ionisation energy of Na is lower than that of Mg.
Na has lower /weaker nuclear attraction for the outer electron than Mg because Mg has more protons with similar shielding (1)
Therefore less energy is required to remove Na’s outer electron / Na’s outer electron is held less strongly (1)
(5 marks) Order the first ionisation energies of Al, Si, P, and S from lowest to highest. Explain your ordering using shielding, , and any relevant electron-arrangement effect.
Correct order: Al < Si < S < P (1)
Across the period, shielding is similar and increases, so IE tends to rise from Al to P (1)
Al lower because its outer electron is in a higher-energy 3p subshell (easier to remove) compared with Si/P where attraction is greater (1)
P higher than S due to half-filled 3p arrangement in P (reduced repulsion / extra stability) (1)
S slightly lower because paired 3p electrons increase repulsion, easing removal despite higher (1)

