AP Syllabus focus: ‘Predict and explain electron affinity and electronegativity trends using Coulomb’s law and electronic structure patterns across the periodic table.’
Electron affinity and electronegativity are related measures of how strongly atoms attract electrons. AP Chemistry emphasizes explaining their periodic trends using electrostatic attraction and valence-shell structure rather than memorising isolated values.
Core ideas and key definitions
Electron affinity (EA)
Electron affinity: The energy change when an electron is added to a gaseous atom to form a gaseous anion; .
EA is often discussed by its sign: a more negative EA means the atom releases more energy upon gaining an electron (electron gain is more energetically favourable).
Electronegativity (EN)
Electronegativity: A relative measure of an atom’s ability to attract shared electrons in a chemical bond.
EN is dimensionless (a scale), so trends matter more than absolute numbers.
Using Coulomb’s law to justify trends
Coulomb’s law connects the strength of attraction to nuclear charge and electron–nucleus distance.
For periodic trends, you apply it qualitatively: stronger attraction tends to make EA more negative and EN higher.
= electrostatic force (newtons, N)
= interacting charges (coulombs, C)
= separation distance (meters, m)
= Coulomb constant
Across a period, effective nuclear charge (often written as ) increases because protons are added while shielding changes only modestly. Down a group, the valence electrons are in higher shells, so increases and shielding increases, weakening attraction.
Electron affinity trends (qualitative)
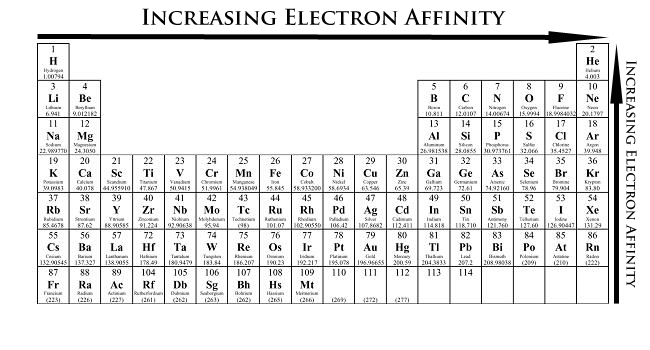
Periodic-table heatmap showing how electron affinity varies by position, with the strongest (most exothermic/most negative) electron affinities concentrated toward the upper-right of the main-group elements. This makes it easier to see why halogens typically have very favorable electron affinities, while several regions (notably parts of Groups 2 and 18) deviate from the simple left-to-right pattern. Source
General trend across a period (left → right)
EA tends to become more negative.
Reasoning:
Increasing strengthens attraction for an added electron.
Valence-shell radius generally decreases, reducing and increasing attraction.
General trend down a group (top → bottom)
EA tends to become less negative (less energy released).
Reasoning:
Added electron enters a higher principal energy level, increasing distance from the nucleus.
Increased shielding by core electrons lowers the net attraction.
Important pattern-based exceptions (what to say, not to calculate)
Noble gases: EA is typically unfavourable (not strongly negative) because adding an electron would require placing it in a new shell, increasing and reducing stabilisation.
Group 2 (alkaline earths): relatively less negative EA because the added electron must enter a higher-energy p subshell after a filled s subshell.
Group 15: relatively less negative EA because adding an electron pairs it within a p subshell, increasing electron–electron repulsion.
Halogens: among the most negative EA values because gaining one electron completes a valence shell with strong nuclear attraction and manageable repulsions.
When comparing two atoms’ EA, support claims with:
relative (periodic position),
relative valence-shell distance ( level),
whether the added electron causes pairing/repulsion in a subshell.
Electronegativity trends (qualitative)
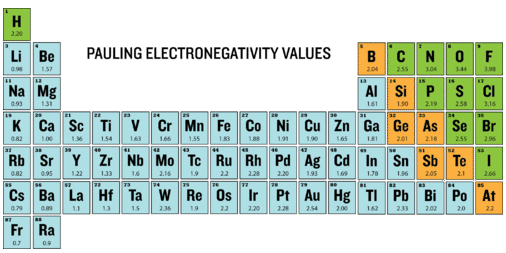
Periodic table annotated with Pauling electronegativity values, highlighting the increase across a period and the decrease down a group. The concentration of high values near fluorine makes the “top-right (excluding noble gases)” trend immediately visible and supports qualitative comparisons between metals and nonmetals. Source
General trend across a period (left → right)
EN increases.
Reasoning:
Higher increases attraction for shared electrons.
Smaller atomic radius reduces , strengthening attraction to bonding electrons.
General trend down a group (top → bottom)
EN decreases.
Reasoning:
Bonding electrons are farther from the nucleus (larger ).
Greater shielding reduces the nucleus’s pull on the bonding pair.
Key comparisons students should justify
Nonmetals generally have higher EN than metals in the same period due to stronger attraction for bonding electrons.
Fluorine is the most electronegative element because it combines high with very small radius.
EN is not meaningfully assigned to noble gases in many contexts because they form few bonds; this reflects “electronegativity as a bonding concept,” not a failure of periodicity.
Linking EA and EN (what is similar and what is different)
Similar:
Both depend on nucleus–electron attraction and therefore reflect , shielding, and distance.
Both generally increase in “electron-attracting ability” across a period and decrease down a group.
Different:
EA is an energy change for forming an anion (isolated species, gas phase).
EN is an atom-in-a-bond property (competition for shared electrons), so it also reflects the bonding environment and the other atom present.
FAQ
If the added electron must enter a new shell or a very unfavourable arrangement, the stabilising attraction is outweighed by energy costs (poor overlap with the nucleus and increased repulsions), making electron gain endothermic.
After forming $X^-(g)$, adding another electron means forcing an electron onto a negatively charged ion. Electrostatic repulsion dominates, so energy must be supplied to form $X^{2-}(g)$.
They are built from different experimental inputs, but all track how strongly nuclei attract bonding electrons. As a result, periodic changes in $Z_\text{eff}$ and radius produce consistent relative ordering.
Fluorine’s added electron enters a very compact $2p$ region where electron–electron repulsion is relatively large. In chlorine, the added electron goes into a larger $3p$ region with less repulsion, so more energy is released.
EN describes attraction for shared electrons, not the number of electrons transferred. Oxidation state depends on electron accounting rules and bonding context, whereas EN is a comparative tendency that influences bond polarity.
Practice Questions
(2 marks) State the trend in electronegativity down Group 17 and explain it using shielding and distance from the nucleus.
EN decreases down the group. (1)
Explanation: increased shielding and/or increased atomic radius (greater distance) reduces attraction for the bonding pair of electrons. (1)
(5 marks) Element A is in Period 2, Group 15 and element B is in Period 2, Group 17. (a) Predict which element has the more negative electron affinity. (1) (b) Explain your choice using Coulomb’s law ideas and electron subshell electron–electron repulsion. (4)
B has the more negative EA. (1) (b)
Across a period increases, strengthening attraction for an added electron (Coulomb’s law: larger effective charge and smaller ). (1)
Group 17 gains an electron to complete the valence shell, giving a strong stabilisation. (1)
Group 15 has a half-filled subshell; adding an electron causes pairing in a orbital. (1)
Pairing increases electron–electron repulsion, making EA less negative for Group 15 than expected. (1)

