AP Syllabus focus: ‘Predict trends in atomic and ionic radii using periodic-table position, the shell model, and qualitative electrostatic reasoning.’
Atomic and ionic size trends come from how electrons are arranged in shells and how strongly the nucleus attracts those electrons. Use periodic-table position to predict relative sizes and justify them with shielding and nuclear attraction.
Atomic radius trends
What “atomic radius” means
Because an atom’s edge is not sharp, atomic radius is defined operationally from measured distances between nuclei in substances.
Atomic radius: A measure of atomic size, commonly taken as half the distance between the nuclei of two identical atoms bonded together (or adjacent in a solid).
Across a period (left → right): radius decreases
From left to right within the same period:
Nuclear charge increases (more protons).
Added electrons go into the same principal shell (same average distance scale).
Shielding changes only slightly because no new inner shell is added. Result: stronger attraction between nucleus and valence electrons pulls the electron cloud in, so atomic radius decreases.
Key periodic-table cue:
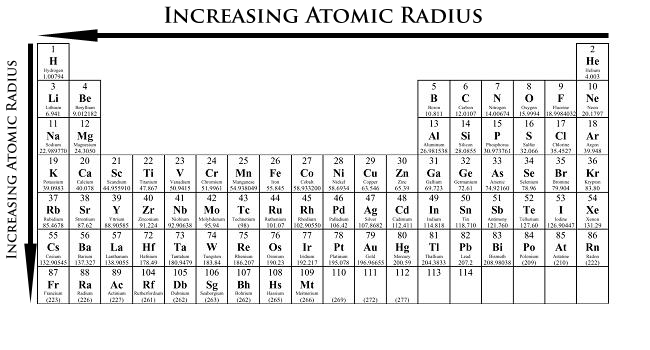
Atomic radius trend on the periodic table. The arrows summarize the standard trend: atomic radius increases down a group (added shells, greater shielding) and increases toward the left across a period (lower effective nuclear attraction on valence electrons). Use this as a quick visual check after you justify a comparison with shell model and qualitative electrostatic reasoning. Source
Elements further to the right (same row) are generally smaller atoms than those on the left.
Down a group (top → bottom): radius increases
From top to bottom within the same group:
Each step adds a new electron shell (higher ).
Valence electrons are, on average, farther from the nucleus.
Inner electrons shield the outer electrons from nuclear attraction. Result: atomic radius increases down a group despite increased nuclear charge, because distance and shielding dominate.
Shell model and qualitative electrostatic reasoning
The shell model (electrons in principal energy levels) supports two size controls:
Distance: electrons in higher shells occupy a larger volume.
Attraction: the nucleus attracts electrons; greater effective pull contracts the radius.
Shielding: Reduction in the attraction between the nucleus and a valence electron due to repulsion from inner (core) electrons.
In qualitative electrostatic terms:
More shielding and greater electron–electron repulsion tend to increase size.
Greater nuclear attraction to valence electrons tends to decrease size.
Ionic radius trends
What “ionic radius” means
An ionic radius reflects the size of an ion in an ionic crystal, inferred from measured distances between ions.
Ionic radius: A measure of ion size based on distances between neighboring ions in an ionic lattice, assigned to cations and anions by a consistent convention.
Cations vs their atoms: cations are smaller
When an atom forms a cation:
Electrons are removed, reducing electron–electron repulsion.
The remaining electrons experience greater attraction per electron.
Often, the outermost shell is partially or completely emptied, decreasing size dramatically. Therefore, cations are smaller than their neutral atoms.
Anions vs their atoms: anions are larger
When an atom forms an anion:
Electrons are added, increasing electron–electron repulsion within the valence shell.
The same nuclear charge must attract more electrons. Therefore, anions are larger than their neutral atoms.
Down a group: ionic radius increases
For ions of the same charge within a group (e.g., to , or to ):
Additional shells are added down the group.
Shielding increases. So ionic radius increases down a group.
Comparing ions with the same number of electrons (isoelectronic reasoning)
Isoelectronic series: more protons → smaller ion
Ions (and sometimes atoms) with the same number of electrons form an isoelectronic series.
Isoelectronic: Species that have the same number of electrons (and therefore similar electron configurations) but different numbers of protons.
Within an isoelectronic series:
Electron count (and shell structure) is constant.
Nuclear charge varies, changing attraction. Therefore:
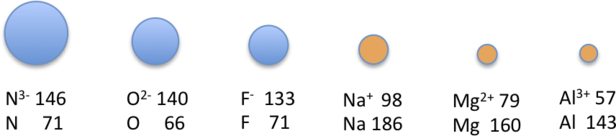
Isoelectronic series size trend (Ne-like, 10 electrons). The ions shown all have the same electron count, so the key variable is nuclear charge: increasing protons strengthens electrostatic attraction and contracts the electron cloud. The diagram makes the qualitative rule explicit: more protons smaller ionic radius within an isoelectronic series. Source
More protons (more positive nucleus) pulls the same electron cloud in more strongly → smaller radius.
Fewer protons → larger radius.
Common AP-level pitfalls to avoid
Do not compare sizes without stating a basis (same period, same group, or isoelectronic).
Remember: forming a cation can remove an entire shell (large size drop), while forming an anion does not add a new shell in the same period.
When explanations sound similar, anchor them to the syllabus language: periodic-table position, shell model, and qualitative electrostatic reasoning.
FAQ
Different radii come from different measured contexts (bonded vs non-bonded contacts). Absolute values change, but the main periodic trends (decrease across, increase down) are typically preserved.
If ion formation removes the last electron(s) from the outer shell, the ion loses an entire shell. The highest occupied shell then has a much smaller average distance from the nucleus.
Use an isoelectronic comparison if possible. If not, compare (i) highest occupied shell ($n$) and (ii) relative nuclear attraction versus shielding, stating which effect dominates.
Anions have extra electrons packed into the same valence shell, increasing electron–electron repulsion, while nearby cations have fewer electrons and may have lost the outer shell entirely.
Ionic radii are inferred from measured internuclear distances in ionic crystals (e.g., from diffraction). A consistent partitioning of the cation–anion distance assigns a radius to each ion.
Practice Questions
(1–3 marks) Arrange , , and in order of increasing radius. Briefly justify.
Correct order: (1 mark)
Justification that cations are smaller due to electron loss/increased attraction (1 mark)
Justification that anions are larger due to electron gain/increased repulsion (1 mark)
(4–6 marks) The ions , , , and are isoelectronic. Rank them from largest to smallest ionic radius and explain using electrostatic reasoning.
Identifies they have the same number of electrons (1 mark)
Correct ranking: (2 marks)
Explanation: increasing nuclear charge across the series increases attraction, decreasing radius (2 marks)
Notes electron cloud/shell structure is constant so proton number is the key variable (1 mark)

