AP Syllabus focus: ‘The periodic table is organized by recurring properties that reflect patterns in ground-state electron configurations and filled or partially filled shells.’
The periodic table is not just a list of elements; it is a map of repeating (periodic) chemical behavior. Its layout comes directly from how electrons occupy energy levels and how those electrons control bonding and reactivity.
Big idea: periodicity comes from electron structure
Chemists organize elements so that similar chemical properties recur at regular intervals as atomic number increases.
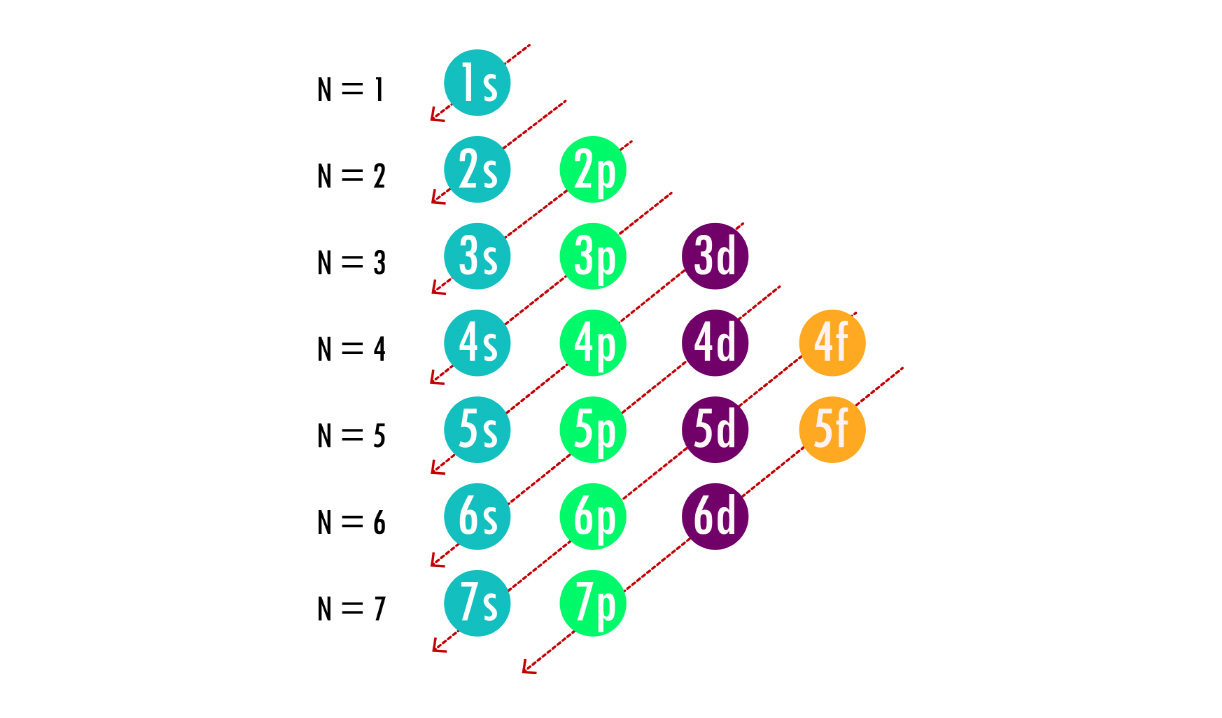
Diagonal-arrow (Aufbau) diagram showing the order in which atomic subshells fill in the ground state (e.g., ). It visually encodes how electron structure changes predictably with increasing atomic number, providing the foundation for periodic recurrence of chemical properties. Source
Those recurring properties arise because electrons fill shells and subshells in predictable patterns, and chemical behavior depends strongly on the outer (highest-energy) electrons.
Periodic law: When elements are arranged by increasing atomic number, their physical and chemical properties repeat periodically due to repeating patterns in electron configuration.
This “repeat” is what makes a single table useful for predicting trends in bonding, formulas of compounds, and reactivity patterns.
Periods and groups: what the rows and columns mean
Periods (rows) reflect filled shells
A new period begins when electrons start occupying a new principal energy level (a new electron shell). Moving left to right across a period:
electrons are added to the same overall shell (for main-group elements)
the valence-shell pattern changes in a regular way, producing a progression of properties from metallic to nonmetallic character
A key organisational idea is that the period number corresponds to the highest occupied principal energy level in the ground state for many elements (especially in the s and p blocks), linking table position to electron arrangement.
Groups (columns) reflect similar valence electron patterns
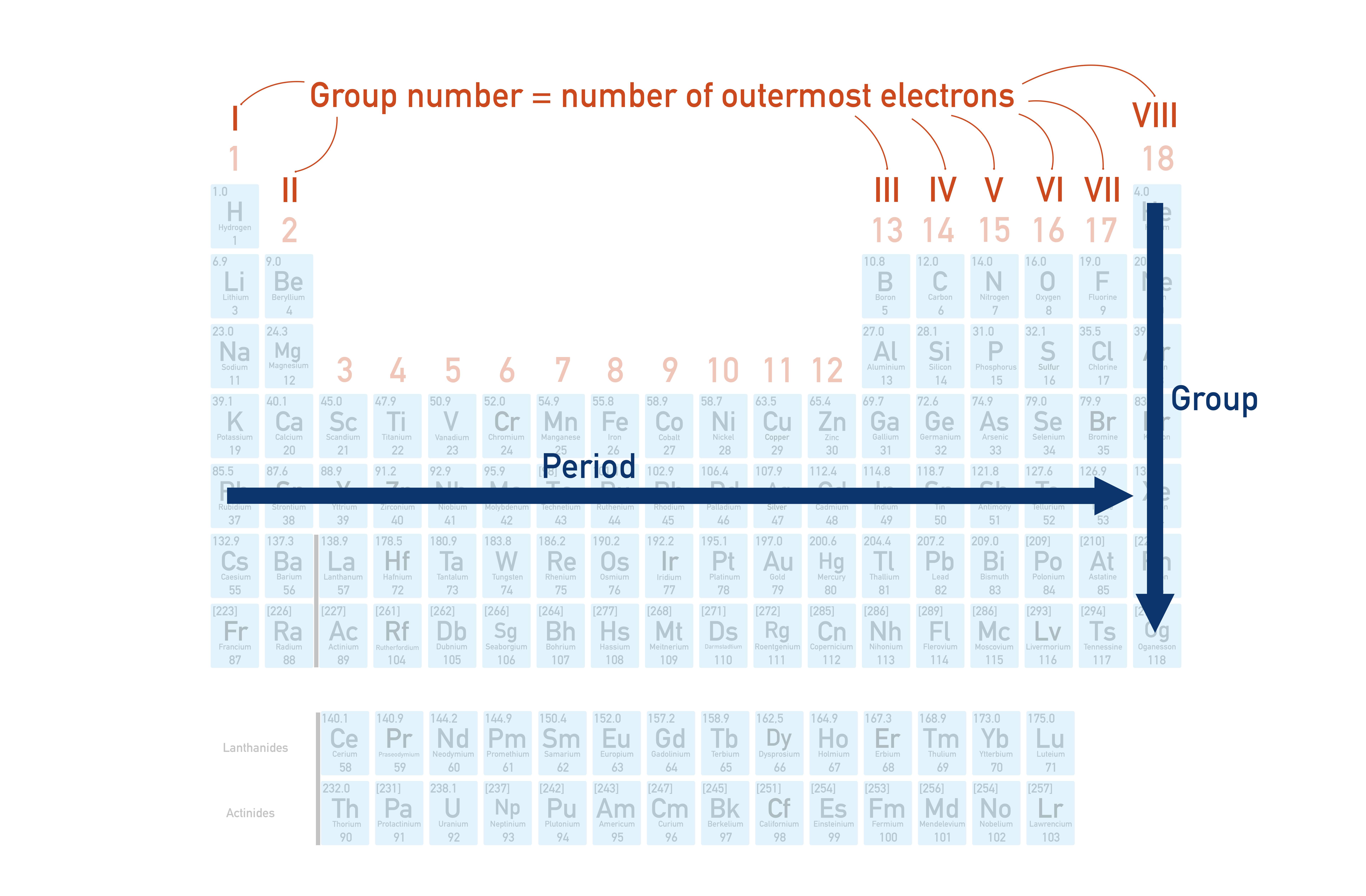
Annotated periodic table highlighting how periods run horizontally and groups run vertically, with an explicit note connecting group position to the number of outer (valence) electrons for main-group elements. This reinforces why elements in the same column share similar valence electron configurations and therefore exhibit similar bonding and reactivity patterns. Source
Elements in the same group are placed together because they share similar valence electron configurations, so they tend to form analogous ions and compounds and show related reactivity.
Valence electrons: Electrons in the outermost occupied shell of an atom; these are most directly involved in bonding and largely determine chemical properties.
For example, main-group elements in a given group typically share the same number of valence electrons, so their bonding preferences and common oxidation states often match.
Blocks: why the table has its characteristic shape
The table’s “blocks” exist because electrons fill specific subshell types in a characteristic order. The block labels tell you which subshell is being filled by the differentiating electron in the ground state.
Block (s, p, d, f): A region of the periodic table defined by the subshell type being filled in the ground-state electron configuration.
The four blocks and what they imply
s-block (Groups 1–2 plus He): filling an s subshell; often features very reactive metals (except He)
p-block (Groups 13–18): filling a p subshell; includes many nonmetals and metalloids; wide variety of bonding
d-block (transition metals): filling a d subshell; often multiple common oxidation states and complex ion formation
f-block (lanthanides/actinides): filling an f subshell; placed separately to keep the table compact while preserving periodic relationships
This block structure ensures that elements with similarly structured outer electron arrangements align in ways that make their recurring properties visually and predictively accessible.
Recurring properties: filled and partially filled shells
The syllabus focus links organisation to ground-state electron configurations and to filled or partially filled shells. Chemically:
Filled shells are especially stable, so elements with filled valence shells cluster into a family with distinctive low reactivity.
Partially filled shells often drive reactivity because atoms tend to gain, lose, or share electrons to reach more stable arrangements.
Families that emerge from repeating valence patterns
Elements at the start of a period tend to have few valence electrons and often behave like metals (electron loss is common).
Elements near the end of a period tend to have many valence electrons and often behave like nonmetals (electron gain or sharing is common).
A repeating sequence of valence configurations across each period recreates recognisable families (columns), which is why the periodic table can predict “like reacts like.”
FAQ
Helium has a filled valence shell (its first shell is complete at 2 electrons), giving it very low reactivity like the noble gases.
Its placement prioritises chemical behaviour (inertness) over block-purity.
They belong in the main table as part of periods 6 and 7, but inserting them inline would make the table extremely wide.
They are separated for compactness while keeping consistent group relationships.
A new period begins when electrons start filling a new principal energy level.
That new shell restarts the pattern of valence configurations, recreating similar families across successive rows.
Atomic number corresponds to proton count and therefore electron count in neutral atoms, which determines ground-state electron configuration.
Atomic mass can vary due to isotopes and does not consistently predict electron structure.
They correspond to filling of the $d$ subshell, which occurs after the $s$ subshell of the same period begins filling.
This produces a central block that bridges the s- and p-block patterns in each applicable period.
Practice Questions
(2 marks) Explain why elements in the same group of the periodic table tend to have similar chemical properties. Refer to ground-state electron configurations.
1 mark: States that elements in the same group have the same (or very similar) valence electron configuration / same number of valence electrons.
1 mark: Links valence electrons to chemical properties (bonding/reactivity) because valence electrons are involved in forming bonds.
(6 marks) Four elements have the following outer (highest-energy) ground-state electron configurations: A: B: C: D:
(a) Identify which letter corresponds to an element in Group 1. (1 mark)
(b) Identify which letter corresponds to a noble gas. (1 mark)
(c) For B and D, state the block (s or p) and justify each choice using the given configurations. (2 marks)
(d) State which pair would be expected to show the most similar chemical behaviour and explain why. (2 marks)
(a) 1 mark: A (has ).
(b) 1 mark: D (has , filled valence shell).
(c) 1 mark: B is p-block because the differentiating electrons are in (configuration ends in ).
(c) 1 mark: D is p-block because the configuration ends in (p subshell filled).
(d) 1 mark: B and D (same group pattern in the sense of both being p-block nonmetals at the end of a period) OR better: A and C (both s-block metals) depending on framing; award for selecting a defensible “most similar” pair.
(d) 1 mark: Explanation links similarity to similar valence-shell structure (same block and/or similar valence electron arrangement leading to similar bonding/reactivity).

