AP Syllabus focus: ‘Coulomb’s law describes the electrostatic force between charged particles and helps explain attraction between the nucleus and electrons.’
Electrostatic forces govern how charged particles interact inside atoms. Coulomb’s law provides a simple, powerful way to connect charge and distance to the strength of attraction or repulsion between nuclei and electrons.
Core Idea: Electrostatic Force in Atoms
Atoms contain a dense, positively charged nucleus (protons) and negatively charged electrons surrounding it. The behavior and energies of electrons are strongly influenced by the electrostatic interaction between these charged particles.
Coulomb’s Law (Qualitative Meaning)
As the distance between charges changes, the force changes dramatically.
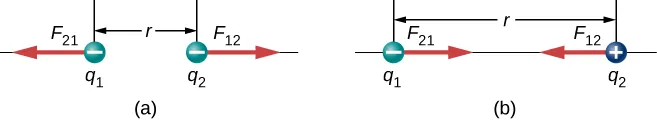
Two point charges separated by distance r exert equal-magnitude forces on each other along the line joining them. The diagram contrasts like-charge repulsion with unlike-charge attraction and makes the force-direction change visually explicit while keeping the geometry simple and labeled. Source
As the magnitude of charge increases, the force increases. Whether the interaction is attractive or repulsive depends on the signs of the charges.
Electrostatic (Coulombic) force: The force of attraction or repulsion between two charged particles due to their electric charges.
In atomic systems:
Opposite charges attract: nucleus (+) and electron (−) experience attraction.
Like charges repel: electron–electron repulsion and proton–proton repulsion exist (though protons are bound in the nucleus by non-electrostatic forces not treated by Coulomb’s law at the AP level).
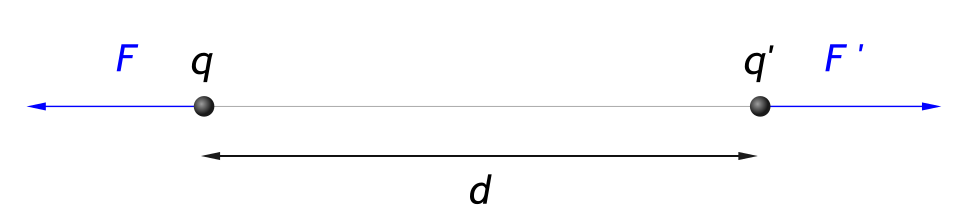
Repulsive interaction between two like charges separated by distance r, with force vectors pointing away from each other. The equal-and-opposite arrows emphasize that each charge experiences the same force magnitude but in opposite directions, consistent with Newton’s third law. Source
Coulomb’s law can be used to compare relative force strengths in different atomic situations (for example, inner vs. outer electrons) without doing detailed calculations.
F=kr2q1q2
F = electrostatic force magnitude, in N
$</p><p>k=Coulomb’sconstant,8.99\times 10^9\ \text{N·m}^2\text{/C}^2</p><p></p><p>q_1,\ q_2=chargesoftheinteractingparticles,inC</p><p></p><p>r=distancebetweenthecentersofthecharges,inm</p></div><p>Thisrelationshipisoftenappliedqualitativelyinchemistrybyfocusingontwobigideas:<strong>chargemagnitude</strong>and<strong>distance</strong>.</p><h2class="editor−heading"id="applying−coulomb−s−law−to−nucleus−electron−attraction"><strong>ApplyingCoulomb’sLawtoNucleus–ElectronAttraction</strong></h2><p>Coulomb’slawhelpsexplainwhyelectronsclosertothenucleusaregenerallyheldmoretightly.</p><h3class="editor−heading"><strong>DistanceDependence(r)</strong></h3><p>Becauseforcescaleswith1/r^2:</p><ul><li><p>Smalldecreasesinelectron–nucleusdistancecancauselargeincreasesinattractiveforce.</p></li><li><p>Electronsin<strong>innershells</strong>experiencestrongerattractiontothenucleusthanelectronsin<strong>outershells</strong>,largelybecausetheyarecloseronaverage.</p></li></ul><p>Thisiswhy:</p><ul><li><p>Innerelectronstendtohave<strong>lowerpotentialenergy</strong>(morestable,moretightlybound).</p></li><li><p>Outerelectronsaregenerallyeasiertoremoveorshareinchemicalprocessesbecausetheyexperienceweakerattraction.</p></li></ul><h3class="editor−heading"><strong>ChargeDependence(q_1 q_2)</strong></h3><p>Inanatom,therelevantchargeinteractionisbetween:</p><ul><li><p>the<strong>nucleus</strong>(netpositivechargethatgrowswithnumberofprotons),and</p></li><li><p>an<strong>electron</strong>(fixedcharge,−1elementarycharge).</p></li></ul><p>Asnuclearpositivechargeincreases,theattractiveforceonelectronscanincrease,butthefulleffectdependsonhowotherelectronsmodifytheinteraction.</p><h2class="editor−heading"id="electron−electron−repulsion−and−shielding−conceptual−link"><strong>Electron–ElectronRepulsionandShielding(ConceptualLink)</strong></h2><p>Electronsdonotonlyfeelattractiontothenucleus;theyalsoexperience<strong>repulsionfromotherelectrons</strong>.Thisrepulsionreduceshowstronglyouterelectronsarepulledinward.</p><p>Keyqualitativeconsequences:</p><ul><li><p>Innerelectronscanpartially“block”nuclearattractionfromreachingouterelectrons,soouterelectronsexperienceareducedpull.</p></li><li><p>Electronsinthesamegeneralregioncanrepeleachother,influencinghowelectrondensityisdistributedandhowstronglyparticularelectronsareheld.</p></li></ul><p>Coulomb’slawunderliesbotheffectsbecauseelectron–electronrepulsionisalsoaninverse−squareelectrostaticinteraction.</p><h2class="editor−heading"id="what−coulomb−s−law−helps−you−explain−ap−level−uses"><strong>WhatCoulomb’sLawHelpsYouExplain(AP−LevelUses)</strong></h2><p>Coulomb’slawisusedasareasoningtooltoconnectatomicstructuretoobservablebehavior:</p><ul><li><p>Why<strong>electronsclosertothenucleus</strong>aregenerallymorestronglyattracted(smallerr$).
Why increasing nuclear charge can increase attraction (larger effective positive pull), especially when distance is similar.
Why electron repulsion matters in determining how strongly outer electrons are held.



