AP Syllabus focus: ‘Electrons occupy shells and subshells. Ground-state electron configurations describe how electrons are distributed among these energy levels.’
Electron arrangement determines many chemical properties. AP Chemistry describes electron locations using energy levels (shells), sublevels (subshells), and orbitals, along with basic filling rules that produce an atom’s lowest-energy (ground-state) electron configuration.
Shells: principal energy levels
Electrons in atoms occupy shells, which are broad energy levels centered around the nucleus. Shell number increases as electrons, on average, occupy regions farther from the nucleus and higher in energy.
Shell (principal energy level): A main electron energy level (often labelled ) containing one or more subshells; higher shells generally correspond to higher energy and larger average distance from the nucleus.
Shells help explain why elements in the same period fill the same overall energy level: moving across a period adds electrons to the same principal shell until it is energetically favourable to begin a new shell.
= maximum number of electrons the shell can hold, in electrons
= shell (principal energy level) number, unitless
This capacity is a useful upper limit, but actual filling depends on subshell energies within and between shells.
Subshells: s, p, d, f
Within each shell, electrons are grouped into subshells (also called sublevels). Subshells differ in shape and energy and are labelled s, p, d, f.
Subshell: A subdivision of a shell containing orbitals of the same type (s, p, d, or f) that share similar energy and shape characteristics.
Key subshell facts used in AP Chemistry:
s subshell: 1 orbital, holds up to 2 electrons
p subshell: 3 orbitals, holds up to 6 electrons
d subshell: 5 orbitals, holds up to 10 electrons
f subshell: 7 orbitals, holds up to 14 electrons
Each shell contains specific subshells:
: s only
: s and p
: s, p, and d
: s, p, d, and f (as applicable)
Subshell labels are combined with the shell number (for example, 2p means the p subshell in the second shell).

This diagonal-rule (Aufbau) chart summarizes the relative filling order of atomic subshells (e.g., ). Following the arrows gives the standard sequence used to build ground-state electron configurations from lowest to higher orbital energy. Source
Orbitals: where electrons are most likely found
A subshell is made of individual orbitals.
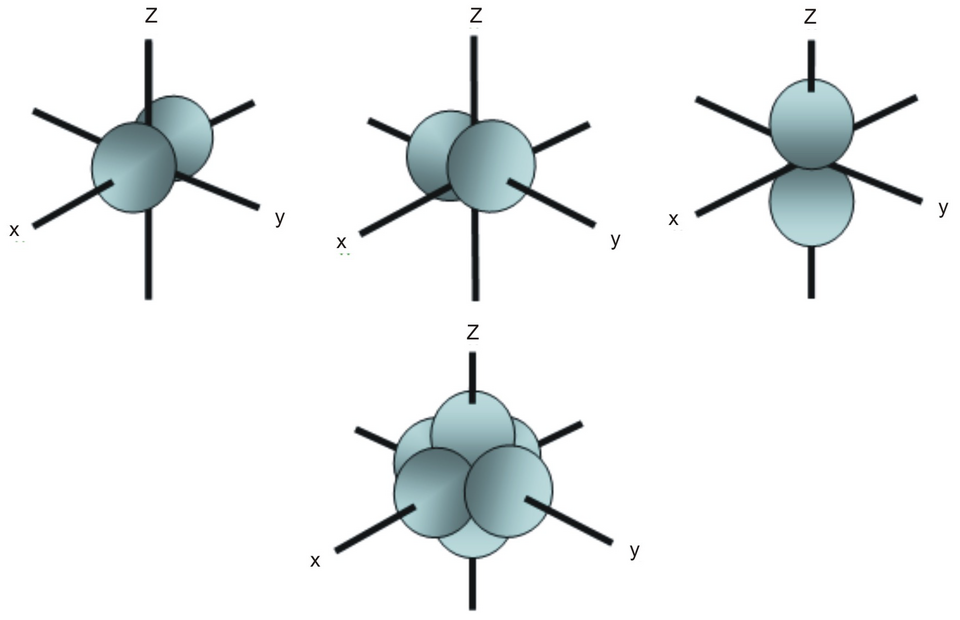
The figure shows the three p orbitals (, , ) as distinct, equal-energy orbitals oriented along perpendicular axes. Seeing them side-by-side helps explain why a p subshell contains three orbitals (and therefore can accommodate up to 6 electrons, two per orbital with opposite spins). Source
An orbital is not a fixed path; it is a region in space where an electron is likely to be found.
Orbital: A region of high probability for finding an electron; each orbital can hold a maximum of two electrons with opposite spins.
Counting orbitals links directly to electron capacity:
s: 1 orbital → max 2 e⁻
p: 3 orbitals → max 6 e⁻
d: 5 orbitals → max 10 e⁻
f: 7 orbitals → max 14 e⁻
Orbital filling rules for ground-state configurations
A ground-state electron configuration describes the arrangement of electrons that minimises the atom’s energy, consistent with electrostatic attractions and electron–electron repulsions.
Ground-state electron configuration: The lowest-energy distribution of an atom’s electrons among shells, subshells, and orbitals.
Two principles govern how electrons fill orbitals within a subshell.
Pauli exclusion principle: An orbital holds at most two electrons, and they must have opposite spins.
In addition, electrons spread out among equal-energy orbitals before pairing.
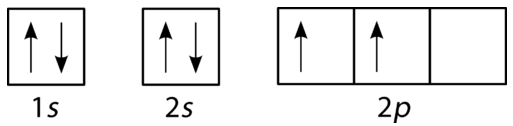
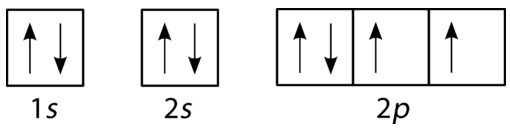
These orbital filling diagrams use boxes (orbitals) and arrows (electron spins) to show how electrons are distributed within subshells. The examples make Hund’s rule visible (single occupancy with parallel spins across degenerate orbitals before pairing) while also enforcing the Pauli exclusion principle (paired electrons in one orbital must have opposite spins). Source
Hund’s rule: For orbitals of the same energy within a subshell (such as the three p orbitals), electrons occupy separate orbitals with parallel spins before any orbital receives a second electron.
Practical implications for AP Chemistry:
Within a subshell, fill one electron per orbital first (Hund), then begin pairing.
Never exceed two electrons per orbital (Pauli).
Electron configurations are commonly written using subshell notation with superscripts (for example, 1s² means two electrons in the 1s orbital).
FAQ
They are degenerate (same energy) in an isolated atom but differ in spatial orientation (often labelled along x, y, z). External fields or bonding environments can break this degeneracy.
Spin is an intrinsic quantum property with two allowed states. In diagrams it is shown as up/down arrows. Opposite spins allow two electrons to share one orbital.
Spreading electrons across orbitals reduces electron–electron repulsion within a subshell. Parallel spins in separate orbitals also correlate with a stabilising exchange effect (treated qualitatively in AP).
The superscript is the total electron count in that subshell, not in a single orbital. For 2p$^5$, five electrons are distributed across three p orbitals following Hund’s rule, then paired as needed.
No. Orbitals represent probability distributions. The “shape” is a map of where an electron is likely to be found, not a trajectory or circular orbit.
Practice Questions
(2 marks) State the maximum number of electrons that can occupy (i) a single orbital and (ii) a p subshell.
(i) 2 electrons (1)
(ii) 6 electrons (1)
(5 marks) An atom has electrons in the 3p subshell. Using Hund’s rule and the Pauli exclusion principle, describe how electrons occupy the three 3p orbitals as electrons are added from 3p to 3p.
Recognises there are three degenerate p orbitals (1)
For 3p, one electron occupies one orbital (unpaired) (1)
For 3p and 3p, electrons occupy separate orbitals before pairing; spins parallel (1)
Pairing begins at 3p after each orbital has one electron (1)
Maximum of two electrons per orbital with opposite spins (Pauli) (1)

