AP Syllabus focus: ‘Represent the ground-state electron configuration of an atom using the Aufbau principle and patterns shown in the periodic table.’
Electron configurations are compact “addresses” for electrons in atoms. Using the Aufbau principle and periodic-table patterns, you can systematically write the ground-state configuration that best predicts many chemical properties.
Core idea: build up electrons in the lowest-energy orbitals first
Aufbau principle: Electrons occupy the available orbitals of lowest energy first, filling higher-energy orbitals only after lower-energy ones are filled.
In AP Chemistry, you use the Aufbau filling order as a reliable algorithm for ground-state configurations of neutral atoms.
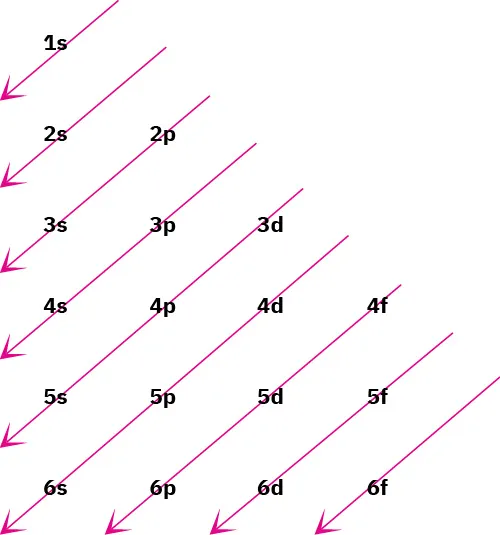
Diagonal-arrow (Aufbau) diagram showing the order in which subshells are filled in ground-state electron configurations. The arrows encode the standard energy ordering (e.g., 4s fills before 3d), which is why configurations are not written in strictly increasing principal energy level. Source
What an electron configuration communicates
An electron configuration shows:
which subshells (s, p, d, f) contain electrons
how many electrons are in each subshell (as superscripts)
the distribution consistent with a lowest-energy (ground-state) arrangement
Electron configuration: A notation that lists the occupied electron subshells of an atom (or ion) and the number of electrons in each subshell.
A correct configuration must account for the element’s total number of electrons (its atomic number for a neutral atom).
The filling order you must know for Aufbau writing
Standard Aufbau sequence (subshell energy order)
Memorise or reproduce this order for filling:
1s
2s
2p, 3s
3p, 4s
3d, 4p
5s
4d, 5p
6s
4f, 5d, 6p
7s
5f, 6d, 7p
Subshell capacities (how many electrons fit)
Use these fixed capacities while filling:
s: 2 electrons
p: 6 electrons
d: 10 electrons
f: 14 electrons
How to write a ground-state electron configuration (process)
Step-by-step algorithm (neutral atoms)
Determine the atom’s total electrons (atomic number).
Fill subshells in the Aufbau sequence.
Do not exceed the subshell capacity (s2, p6, d10, f14).
Stop when all electrons have been placed.
Check your work by summing superscripts to ensure the correct total electron count.
Notation rules and conventions
Write subshells with principal energy level and letter (for example, 3p), followed by a superscript electron count (for example, 3p⁶).
Keep the order consistent with the Aufbau sequence (not strictly increasing principal energy level).
A filled subshell is especially stable and is written at full capacity (for example, 2p⁶, 3d¹⁰).
Using periodic-table patterns to write configurations faster
The periodic table is organised into blocks that match the type of subshell being filled by the last electron added:
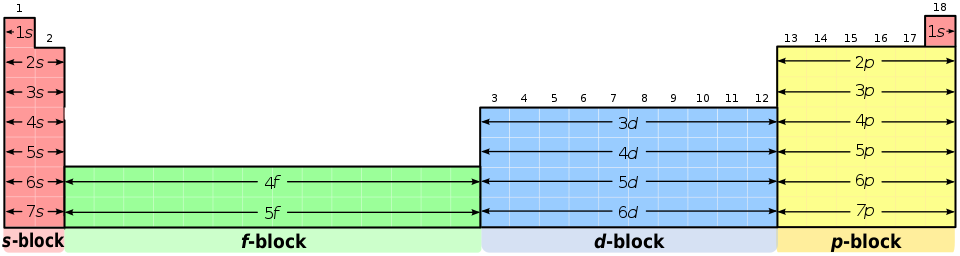
Periodic table colored by subshell block (s, p, d, f), showing how the table’s layout mirrors electron filling by subshell type. This makes it easier to predict the ending of a ground-state configuration (e.g., s-block ends in or ) from an element’s position. Source
s-block: Groups 1–2 (plus He) → ends in ns¹ or ns²
p-block: Groups 13–18 → ends in np¹ through np⁶
d-block: Transition metals → ends in (n−1)d¹ through (n−1)d¹⁰ (with ns electrons included earlier in the writing)
f-block: Lanthanides/actinides → ends in (n−2)f¹ through (n−2)f¹⁴
Noble-gas shorthand (condensed configurations)
To simplify long configurations, replace the core electrons with the preceding noble gas in brackets:
Choose the noble gas at the end of the previous period.
Write the remaining subshells after the bracket in Aufbau order.
This condensed form highlights valence-region electrons while keeping the configuration readable.
Internal consistency checks using the periodic table
The period number often matches the highest principal energy level (n) present in the configuration (especially for s and p blocks).
The group (main-group elements) is consistent with the number of electrons in the outer s and p subshells (for example, ns²np⁵ for halogens).
The block tells you the subshell type of the last-filled electrons, helping you avoid misplaced d or f electrons.
FAQ
Because, in many multi-electron atoms, the $4s$ orbital is slightly lower in energy than $3d$ before electrons occupy the $3d$ subshell.
Energy ordering reflects electron–electron repulsions as well as nucleus–electron attraction.
Yes. An excited-state configuration shows an electron promoted to a higher-energy subshell than predicted by Aufbau filling.
Excited states are not the lowest-energy arrangement, so they are not “ground-state” configurations.
Look at the last subshell written:
ends in $s$ → s-block
ends in $p$ → p-block
ends in $d$ → d-block
ends in $f$ → f-block
It compresses the inner (core) electrons into a bracketed label, keeping focus on the outer electrons that most influence bonding and periodic trends.
Only if you do not track the total electron count. Always sum superscripts until they equal the atom’s total electrons; then stop immediately.
Practice Questions
(2 marks) Write the full ground-state electron configuration of oxygen, O.
1 mark: correct subshell order shown (, , ).
1 mark: correct electron counts ().
(5 marks) Without using a periodic table, write the full ground-state electron configuration of bromine, Br, and also write its noble-gas shorthand.
1 mark: correct filling order through is followed (includes before , and before ).
2 marks: correct full configuration ().
1 mark: correct noble gas core identified ().
1 mark: correct shorthand continuation ().

