AP Syllabus focus: ‘Ionization energy can be estimated qualitatively using Coulomb’s law; it depends on distance from the nucleus and the effective (shielded) nuclear charge.’
Ionization energy reflects how strongly an atom’s nucleus attracts its electrons. In AP Chemistry, you explain ionization energy patterns qualitatively using electrostatic ideas: distance from the nucleus, shielding by other electrons, and effective nuclear charge.
Ionization energy as an electrostatic concept
Ionization energy (IE): The energy required to remove an electron from a gaseous atom (or ion); the first ionization energy removes the outermost electron from a neutral gaseous atom.
Ionization requires overcoming the electrostatic attraction between the negatively charged electron and the positively charged nucleus. Anything that increases attraction increases IE; anything that decreases attraction lowers IE.
Coulomb’s law and nuclear attraction
Electrostatic attraction can be discussed using Coulomb’s law: stronger charges and shorter distances produce stronger attraction, so electrons are harder to remove.
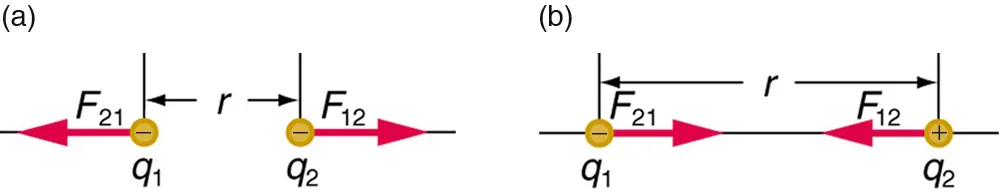
This diagram visualizes Coulomb’s law by comparing the direction of electrostatic forces for like charges (repulsion) versus unlike charges (attraction) at separation distance . It reinforces that the interaction strength depends on charge magnitude and decreases rapidly as increases (inverse-square dependence). Source
= magnitude of electrostatic force (N)
= Coulomb’s constant
= charges of the interacting particles (C)
= distance between charges (m)
In atoms, the relevant ideas are qualitative:
Smaller (electron closer to nucleus) → stronger attraction → higher IE.
Larger nuclear charge (more protons) → stronger attraction → higher IE.
Electron–electron repulsions and shielding reduce how much nuclear charge an outer electron “feels.”
Shielding and effective nuclear charge
Shielding: The reduction in nuclear attraction experienced by an electron due to repulsion from other electrons, especially core electrons between the nucleus and the valence electrons.
Core electrons block some of the nucleus’s pull on outer electrons.
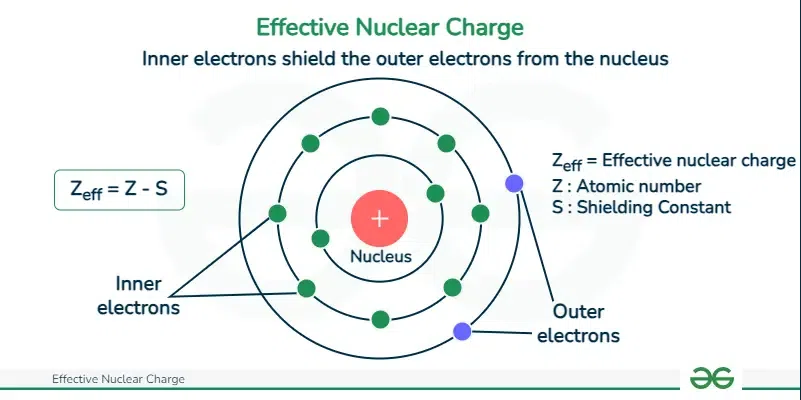
This diagram shows how inner (core) electrons reduce the attractive pull felt by outer (valence) electrons, illustrating the shielding concept. It also connects the picture to the qualitative model of effective nuclear charge using , where shielding lowers the net nuclear pull on a valence electron. Source
As shielding increases, the valence electrons are held less tightly, lowering IE.
A key related idea is the net positive pull felt by a particular electron.
Effective nuclear charge (): The net positive charge experienced by an electron after accounting for shielding by other electrons; higher means stronger attraction to the nucleus.
What increases or decreases (qualitatively)
Increasing number of protons increases nuclear charge and tends to increase for electrons in the same general region of the atom.
Increasing shielding (more inner electrons) decreases felt by valence electrons.
Poor shielding by electrons in the same shell means can still rise even when electrons are added, because added protons may “win” over added repulsions.
Linking IE to distance and (AP-level reasoning)
To estimate relative ionization energies qualitatively, combine two questions:
How far is the electron from the nucleus? (Energy level and overall size of the electron cloud)
How strong is the attraction? (Set mainly by )
Use these cause-and-effect statements:
If a valence electron is in a higher principal energy level, it is on average farther from the nucleus (larger ) and more shielded → lower IE.
If a valence electron experiences a higher , attraction increases even at similar distance → higher IE.
When comparing different electrons within an atom, electrons that are closer in (less distance, typically more penetrating) are harder to remove than those farther out, consistent with stronger Coulombic attraction.
Common reasoning pitfalls to avoid
Shielding is dominated by core electrons; electrons in the same outer shell shield each other only imperfectly.
Ionization energy is about removing an electron from a gaseous species; bonding or phase effects are not part of the concept in this context.
Statements like “more electrons means lower IE” are incomplete; the correct focus is the balance of distance, shielding, and .
FAQ
Penetration describes how much time an electron spends close to the nucleus.
More penetrating electrons experience less shielding and a larger $Z_\mathrm{eff}$.
Less penetrating electrons are kept farther out and feel reduced nuclear attraction.
This helps explain why electrons in different subshells can be held with different strengths even within the same principal shell.
Electrons in the same shell are often at similar average distances from the nucleus, so they do not consistently sit “between” the nucleus and one another.
As a result, same-shell repulsions reduce attraction somewhat, but they do not block the nucleus as effectively as core electrons do.
$Z_\mathrm{eff}$ is mainly a conceptual tool at AP level.
More advanced models can estimate it from experimental data or calculations, but for AP Chemistry you use it qualitatively to justify stronger or weaker nuclear attraction.
Shielding refers specifically to how inner electrons reduce the nucleus’s pull on an outer electron.
Electron–electron repulsion is the broader idea that electrons repel each other; shielding is the part of that repulsion that reduces the effective attraction to the nucleus.
Using gaseous species removes complications from intermolecular forces and lattice interactions.
It ensures the energy change is primarily due to overcoming nucleus–electron attraction (and the internal electron repulsions), making comparisons between elements meaningful.
Practice Questions
(2 marks) Explain, using Coulomb’s law ideas, why an electron closer to the nucleus generally requires more energy to remove.
Mentions smaller distance increases electrostatic attraction/force according to Coulomb’s law. (1)
Links stronger attraction to higher ionisation energy (more energy needed to remove the electron). (1)
(5 marks) An atom has a valence electron that is relatively easy to remove. Using the ideas of shielding and effective nuclear charge, explain two distinct reasons why this could occur, and link each reason to ionisation energy.
Defines or correctly describes shielding as reduction in nuclear attraction due to inner electrons. (1)
Reason 1: increased shielding (more core electrons between nucleus and valence electron) lowers attraction and lowers IE. (1)
Defines or correctly describes effective nuclear charge as net positive charge felt after shielding. (1)
Reason 2: lower (e.g., less nuclear pull felt by valence electron) lowers attraction and lowers IE. (1)
Clear linkage for both reasons to “easier to remove” / lower ionisation energy. (1)

