AP Syllabus focus: ‘Inner electrons are core electrons, while outer electrons are valence electrons; electron configurations distinguish these and help predict chemical behavior.’
Chemists separate electrons into inner and outer sets because they play different roles in attraction to the nucleus and in reactions. Electron configurations make this separation explicit and help you anticipate how an atom will behave chemically.
Core vs Valence Electrons: The Big Idea
Atoms contain electrons in energy levels (shells) and subshells. For most chemical purposes, only the outermost electrons participate directly in forming compounds or reacting, while inner electrons largely remain unchanged.
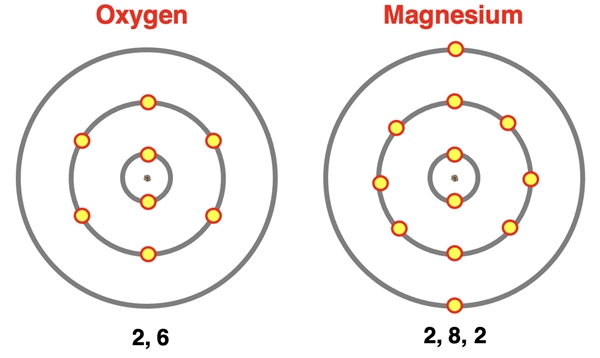
Electron shell diagrams for oxygen and magnesium show electrons grouped by principal energy level, making the “valence shell” easy to spot as the outermost ring. The final shell count (e.g., 2,6 for O and 2,8,2 for Mg) highlights how core electrons remain in inner shells while valence electrons occupy the highest level and govern typical bonding patterns. Source
Core electrons
Core electrons are the inner electrons that fill lower-energy shells and are typically not involved in bonding or gained/lost in ordinary chemical reactions.
Core electrons: Electrons in inner (lower-energy) shells that generally do not participate directly in chemical bonding and remain with the atom during typical reactions.
Key implications of core electrons:
They contribute to shielding: inner electrons reduce the full nuclear attraction felt by outer electrons.
They help determine the atom’s size and the strength with which the nucleus holds valence electrons, indirectly influencing reactivity.
They are usually unchanged when atoms form ions or molecules (in AP Chemistry contexts).
Valence electrons
Valence electrons are the outermost electrons, especially those in the highest principal energy level, that most strongly control chemical behavior.
Valence electrons: Electrons in the outermost occupied energy level of an atom that are most available for bonding and for being removed or added during chemical change.
Valence electrons matter because:
They are farthest from the nucleus on average, so they are held less tightly than core electrons.
They largely determine bonding capacity (how many bonds an atom tends to form) and common reaction patterns.
They explain similarities in chemical behavior among elements with similar outer electron structures.
Using Electron Configurations to Identify Core and Valence Electrons
An electron configuration lists how electrons occupy orbitals (for example, ). To separate core vs valence electrons, focus on which electrons are in the highest numbered shell (largest principal quantum number, ).
A practical procedure (AP-friendly)
Write or read the ground-state electron configuration.
Identify the highest value of present.
Electrons in that highest- shell are the valence electrons.
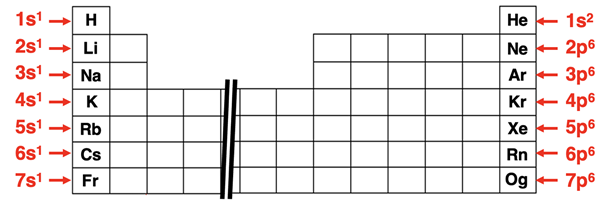
A comparison table of electron configurations highlights valence electrons in red and lists the resulting valence-electron count for several main-group elements. This makes the connection between configuration notation (e.g., in the highest shell) and chemical reactivity patterns explicit and easy to check at a glance. Source
All electrons in lower- shells are core electrons.
Why this works for predicting chemical behavior
Elements with the same number of valence electrons often show similar reactivity patterns because they can reach more stable electron arrangements through similar changes involving those outer electrons.
Changes that happen in reactions (bond formation, ion formation) typically involve rearranging, sharing, gaining, or losing valence electrons, not core electrons.
Core electrons, by shielding, influence how strongly valence electrons are attracted to the nucleus; that influences how readily valence electrons can participate in chemical change.
Common Clarifications Students Need
“Outer” means energy level, not just “last written”
Electron configurations are written in an order that reflects subshell energies, but valence is tied to the outermost shell (highest ). In many main-group cases, the last subshell written is also in the outermost shell, but you should still verify using .
Valence electrons are the “reactive” electrons
In typical chemical processes:
Breaking/forming bonds involves valence electrons.
Spectator inner electrons remain associated with the nucleus and do not need to be tracked explicitly in most reaction reasoning.
Chemical behavior links to valence patterns
Electron configurations let you connect:
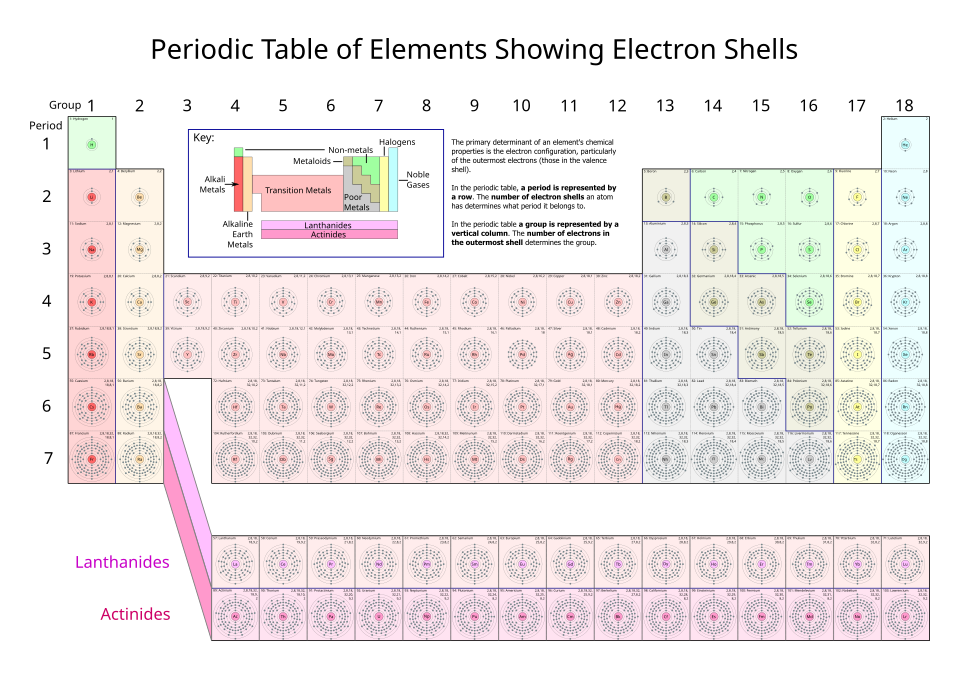
A periodic table annotated with the number of electrons in each shell for every element, reinforcing the idea that shells correspond to principal energy levels (). Reading the last shell entry for an element provides a quick visual cue for the valence-shell population that underlies group-level similarities in chemical behavior. Source
Valence electron count → typical bonding patterns and reactivity tendencies
Core electron shielding → how strongly the nucleus holds the valence electrons (an underlying reason different atoms react differently)
FAQ
Transition metals can involve both $ns$ and $(n-1)d$ electrons in bonding.
Which electrons count as “valence” can depend on context (bonding vs ion formation), so definitions are less straightforward than for main-group elements.
No. Valence electrons are electrons in the outer shell; oxidation state is a bookkeeping number describing electron ownership in compounds.
An element’s valence electron count constrains possible oxidation states but does not equal them.
Core electrons contribute to shielding, changing the attraction between the nucleus and valence electrons.
This alters how tightly valence electrons are held, which can influence how readily bonds form or electrons are removed.
Forming ions typically changes the number of electrons in the outermost shell first.
After ion formation, the “valence shell” can shift because the highest occupied $n$ may change.
Yes. They have valence electrons, but their valence shell is typically full, making them energetically disinclined to react.
“Unreactive” reflects stability of the valence arrangement, not absence of valence electrons.
Practice Questions
(2 marks) Define core electrons and valence electrons, and state which set primarily determines chemical behaviour.
Core electrons defined as inner/low-energy-shell electrons not usually involved in bonding (1)
Valence electrons defined as outermost-shell electrons involved in bonding/chemical change; valence primarily determines chemical behaviour (1)
(5 marks) An atom has the electron configuration . (a) Identify the number of valence electrons. (1) (b) Identify the number of core electrons. (1) (c) Explain, using core vs valence electrons, why the electron configuration helps predict chemical behaviour. (3)
(a) Valence electrons are those with highest : gives 6 (1)
(b) Core electrons are remaining electrons: gives 10 (1)
(c) Any three:
Chemical reactions/bonding mainly involve rearrangement/loss/gain/sharing of valence electrons (1)
Core electrons generally remain unchanged and mainly provide shielding (1)
Knowing the number/arrangement of outer electrons indicates likely reactivity patterns and similarities to atoms with similar valence structure (1)

