AP Syllabus focus: ‘Represent the ground-state electron configuration of common ions by adding or removing electrons from the appropriate energy levels and subshells.’
Ions form when atoms gain or lose electrons. Because chemistry depends strongly on the outer-electron arrangement, you must be able to write ground-state electron configurations for common ions accurately and consistently.
Key idea: electron configurations change by electron count
When forming ions, the number of protons (the element identity) stays the same, while the number of electrons changes. The goal is to write the most stable (lowest-energy) arrangement for the new electron total.
Ion: An atom (or group of atoms) with a net electric charge due to gaining or losing electrons.
A cation is positively charged (lost electrons), and an anion is negatively charged (gained electrons).
Procedure for writing ion electron configurations
1) Start from the neutral atom
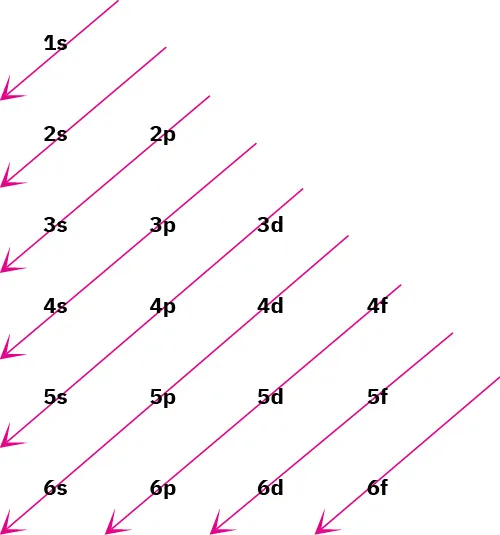
Aufbau (diagonal) filling-order diagram showing the sequence in which subshells are filled in ground-state electron configurations (e.g., 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p). The diagonal arrows provide a fast visual method for determining which subshell is lowest in energy and therefore filled next. Source
Write the neutral atom’s ground-state electron configuration using subshell notation (for example, 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p). Keep track of which subshells contain valence electrons versus inner electrons, since ion formation typically changes the outermost electrons first.
2) Determine the ion’s electron total
Use charge to decide how many electrons to add or remove.
= ionic charge (in units of elementary charge)
= number of protons (atomic number, no unit)
= number of electrons (count, no unit)
This relationship helps you connect the symbol charge (like 2+ or 1−) to the electron count you must place into subshells.
3) For anions: add electrons to the lowest available energy subshell
When forming anions, you add electrons following the usual orbital filling order (lowest energy first) into the existing valence shell.
Add electrons to the current outer subshells (often p subshells for main-group elements).
Stop when the electron count matches the ion’s charge.
Many common anions reach a noble-gas configuration, which is especially stable.
4) For cations: remove electrons from the highest principal energy level (largest n)
When forming cations, remove electrons from the outermost shell first (highest n), not necessarily from the subshell written last in a simple list.
For main-group cations, remove electrons from the largest n level (often from s or p in the valence shell).
Remove one electron per +1 charge.
5) Special rule for transition metals: remove ns before (n−1)d
For many transition metals, the neutral atom fills the ns subshell before the (n−1)d subshell, but cation formation removes ns electrons first.
Example pattern (no calculation): electrons are typically removed from 4s before 3d.
After removing ns electrons, additional electrons (for higher charges) come from the (n−1)d subshell.
This “appropriate subshell” rule is a frequent source of mistakes: focus on the highest n electrons for removal.
Writing conventions you must follow
Subshell notation and exponents
Electron configurations are written with subshell labels and electron counts as superscripts (often typed with caret notation).
Use format like: 3p^6 (meaning 6 electrons in the 3p subshell).
Keep subshell capacities in mind: s holds 2, p holds 6, d holds 10.

Generalized atomic-orbital energy diagram (not to scale) showing that subshell energies increase overall with principal quantum number but also depend on subshell type ( within a shell). The figure highlights common overlaps (e.g., 4s vs. 3d) and summarizes maximum electron capacities for each subshell (2, 6, 10, 14), which is essential for writing correct configurations. Source
Noble-gas shorthand (if used)
You may represent inner (core) electrons with a noble-gas symbol in brackets, then show only the electrons beyond that core.
This is especially helpful for ions, because ion formation usually changes only the outer part of the configuration.
Ensure the bracketed core corresponds to the correct number of electrons for the ion’s remaining inner shells.
Common pitfalls (and how to avoid them)
Removing from the wrong subshell: for cations, remove electrons with the largest n first; for transition metals, remove ns before (n−1)d.
Forgetting the ion charge changes electron count: the element’s identity is fixed by protons, but the configuration must match the ion’s electrons.
Mixing up stability with charge: not every ion becomes a noble-gas configuration, especially for many transition-metal ions; still write the correct ground-state arrangement for the electron total.
FAQ
After $3d$ begins filling, the $3d$ electrons shield and the $4s$ electrons become higher in energy and more exposed, so they are lost first.
Remove/add electrons one at a time using the same rule each step. For high-charge cations, continue removing from the next highest-energy occupied subshell after the outer shell is emptied.
Yes, if the core is correct and the remaining subshell electrons match the ion’s electron count. Use the nearest preceding noble gas for the ion’s electron total, not the neutral atom’s.
Isoelectronic species have the same number of electrons, so they share the same electron configuration. This can help you quickly check whether your electron count and subshell filling are consistent.
Yes. Many ions (especially transition-metal ions) still have partially filled subshells, so unpaired electrons can remain even in the ground state.
Practice Questions
(2 marks) Write the ground-state electron configuration of .
Correct electron total (10 electrons) (1)
Correct configuration: (1)
(5 marks) A neutral iron atom has electrons in the and subshells. Write the ground-state electron configuration of and justify which subshell electrons are removed.
States that electrons are removed to form a cation (1)
Identifies removal from highest principal energy level first (1)
States electrons are removed before for transition metals (1)
Gives correct configuration: (or full equivalent) (2)

