Water as the medium for life
Life first originated in water.
Water remains the medium in which most processes of life occur.
Water is therefore essential as the medium for cell activity, metabolism, and transport.
Polarity and hydrogen bonding
A water molecule has polar covalent bonds because electrons are unequally shared.
Oxygen is partially negative (δ−) and each hydrogen is partially positive (δ+).
The attraction between the δ+ hydrogen of one molecule and the δ− oxygen of another forms a hydrogen bond.
Hydrogen bonding is the basis of water’s key biological properties.
Cohesion, surface tension and biological importance
Cohesion = attraction between water molecules due to hydrogen bonding.
Cohesion helps maintain a continuous column of water in xylem.
Water can be transported under tension in plants.
Surface tension is caused by cohesion at the water surface.
This allows water surfaces to be used as habitats by some organisms.
Adhesion and capillary action
Adhesion = attraction between water and polar or charged materials.
Adhesion helps water move by capillary action.
Important examples are soil and plant cell walls.
Adhesion + cohesion together help pull water upward through plant tissues.
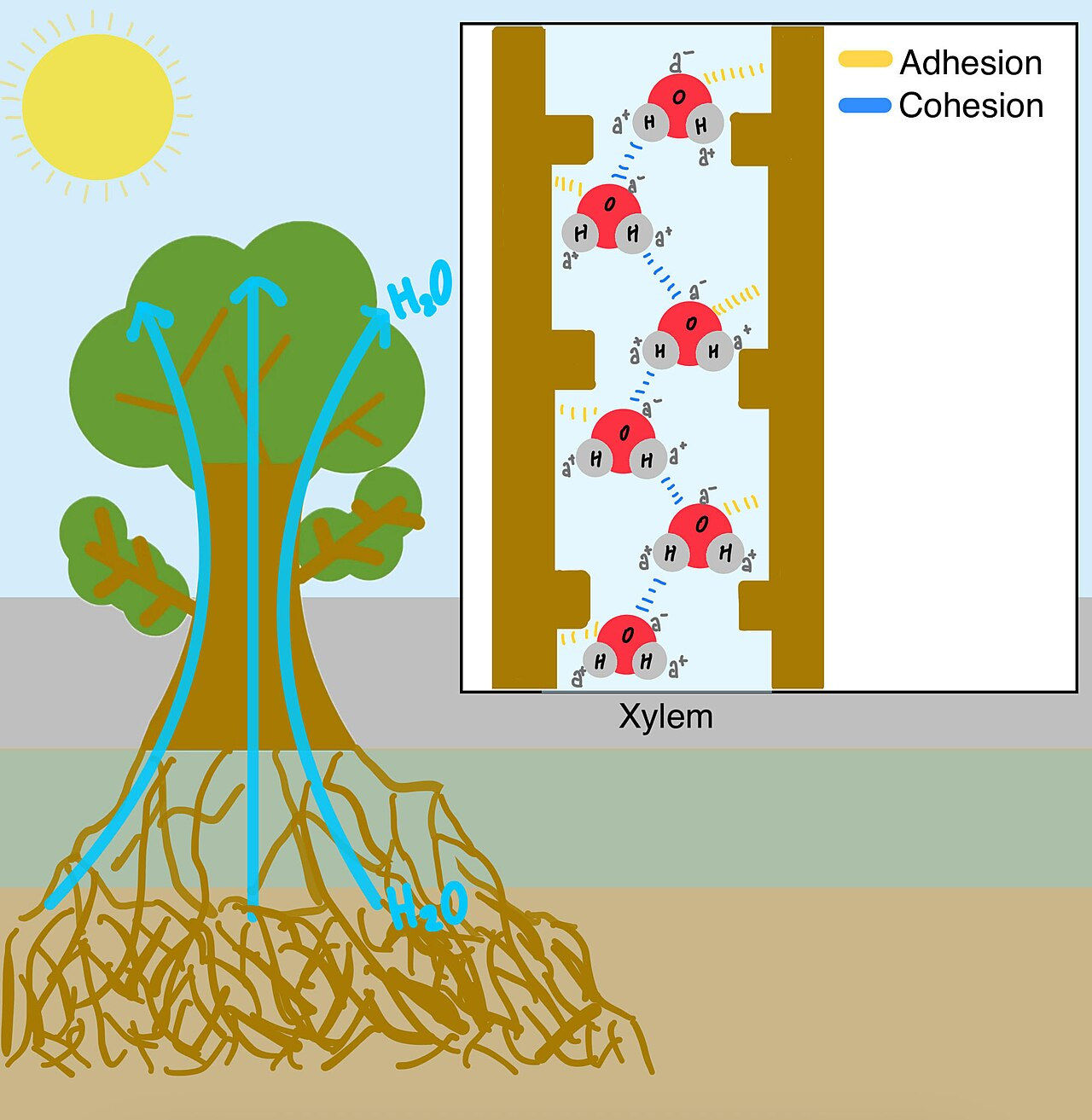
This diagram shows how cohesion between water molecules and adhesion to the xylem wall help move water upward. It is directly useful for explaining capillary action and xylem transport under tension. Source
Solvent properties and role in metabolism
Water is a very effective solvent for many hydrophilic molecules.
This makes water a suitable medium for metabolism and for transport in plants and animals.
Most enzymes catalyse reactions in aqueous solution.
Some molecules in cells must remain hydrophobic and insoluble, and their function depends on this.
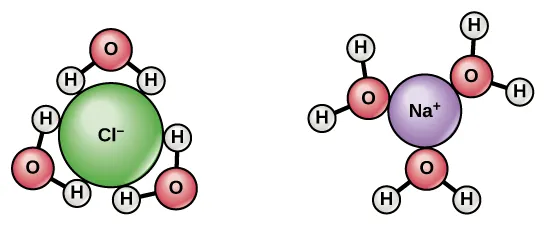
This image shows how water’s polarity allows it to surround and separate charged particles. It explains why water is such an effective solvent for many biologically important substances. Source
Physical properties of water in aquatic habitats
Buoyancy supports organisms in water and reduces the effect of gravity.
Viscosity is greater than in air, so movement through water has more resistance.
Thermal conductivity is greater than in air, so aquatic organisms can lose heat more quickly.
Specific heat capacity is high, so water temperature changes slowly and aquatic habitats are more thermally stable.
In exams, be ready to contrast water with air and relate this to organisms such as the black-throated loon and the ringed seal.
Checklist: can you do this?
Draw or recognize two or more water molecules showing δ+, δ−, and hydrogen bonds.
Explain how polarity leads to hydrogen bonding.
Explain and apply the link between hydrogen bonding and cohesion, surface tension, adhesion, and capillary action.
Interpret why water is a suitable medium for metabolism and transport.
Distinguish between hydrophilic substances that dissolve in water and hydrophobic substances that do not.
Compare water with air for buoyancy, viscosity, thermal conductivity, and specific heat capacity.
Apply these properties to biological examples such as xylem transport, soil water movement, and water-surface habitats.
HL only: Origin of water on Earth
Water on Earth is thought to have an extraplanetary origin.
The syllabus limits this hypothesis to delivery by asteroids.
Water was retained because Earth had enough gravity to hold it, and temperatures became low enough for water to condense.
The long-term abundance of water on Earth allowed life to evolve.
HL only: Water and the search for extraterrestrial life
The search for extraterrestrial life is closely linked to the presence of water.
A key idea is the Goldilocks zone.
This is the region around a star where conditions are not too hot and not too cold, so liquid water could exist.
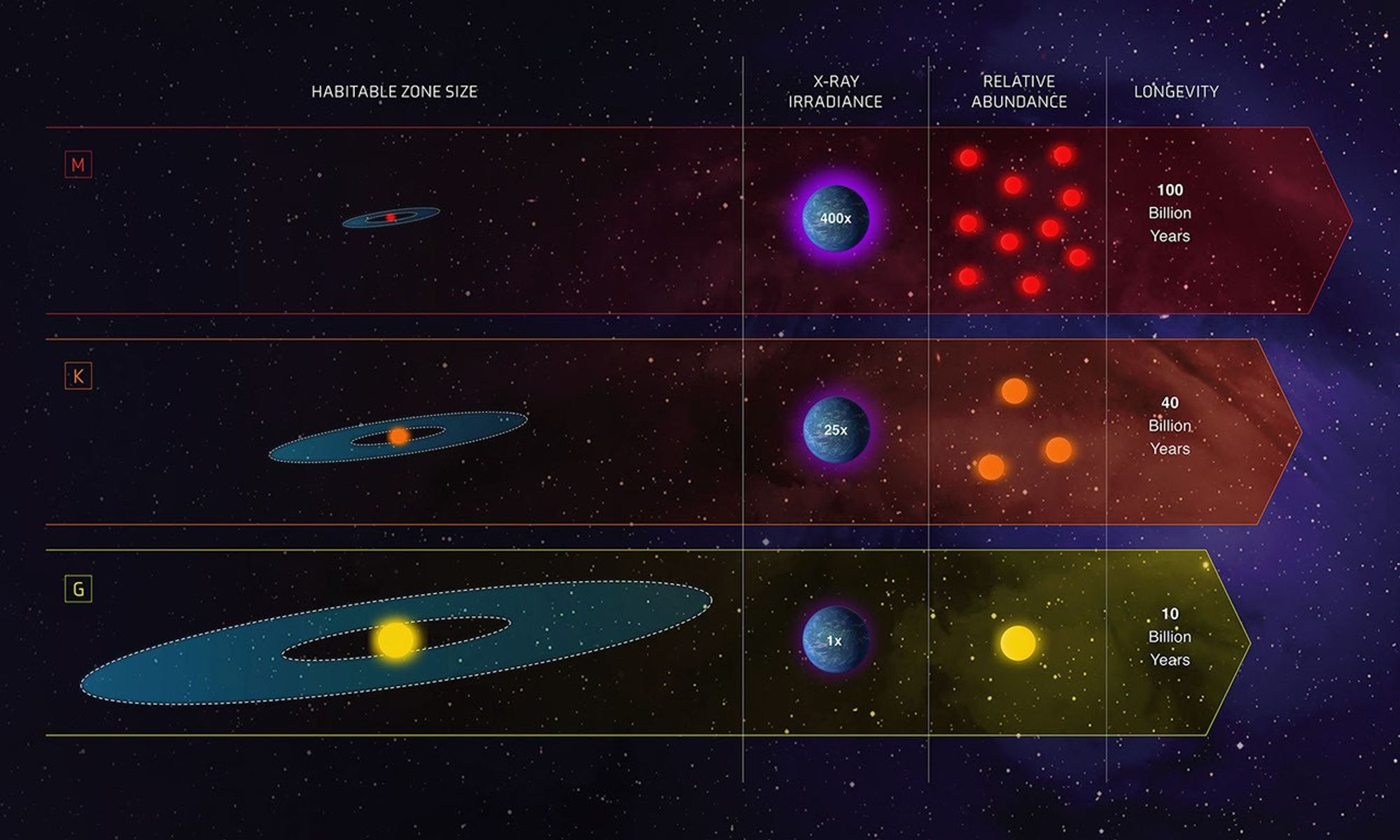
This infographic shows that planets are most promising for life when they orbit in a region where liquid water could exist. It directly supports the HL idea linking water to the search for extraterrestrial life. Source
High-yield link chain
Polar covalent bonds → partial charges → hydrogen bonds.
Hydrogen bonds → cohesion, adhesion, surface tension, solvent properties.
These properties explain water’s roles in metabolism, transport, plant water movement, surface habitats, and aquatic environments.

Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.
Shubhi is a seasoned educational specialist with a sharp focus on IB, A-level, GCSE, AP, and MCAT sciences. With 6+ years of expertise, she excels in advanced curriculum guidance and creating precise educational resources, ensuring expert instruction and deep student comprehension of complex science concepts.